

Bohr Models and Valence Electrons
Presentation
•
Chemistry
•
8th - 11th Grade
•
Hard
Joseph Anderson
FREE Resource
9 Slides • 11 Questions
1
Bohr Models + Ion Formation

2
What we will learn
current atomic model recap
electron shells/configuration
drawing bohr models
ions and ion formation
3
Atomic Structure
Protons + neutrons = nucleus
Electrons = in orbital shells
These are the shells we will be learning about, and the distribution of the electrons in them

4
Electron Shells
Shells around the nucleus can hold certain amounts of electrons (energy)
1st shell = 2 e
2nd shell = 8 e
3rd shell = 8 e
can hold up to 18, but for our purposes we will cap it at 8
Most outer shell = the valence shell --> where it can gain or lose electrons
Valence shell needs to be full for an atom to be considered stable

5
Drawing a Bohr Diagram
Determine numbers of subatomic particles
Draw the nucleus
Draw the 1st shell
Draw subsequent shells (like a clock) until all electrons are shown

6
Multiple Choice

What is the atomic number of this element?
17
18
35
35.453
7
Multiple Choice
8
Multiple Choice
How many electrons can fit on the 1st energy level (orbital) for any Bohr Model?
2
6
8
10
9
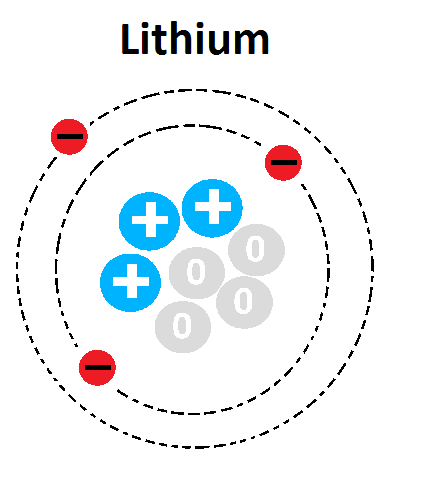
Multiple Choice

What element is represented in this Bohr Model?
Carbon
Hydrogen
Aluminum
Lithium
10
Ion Formation
an ion is a charged particle Or an atom that has gained or lost electrons
positive ions = cations = LOST an electron
negative ions = anions = GAINED an electron
this loss or gain happens to stabilise an atom (give it a full outer shell)

11

12
Naming Ions
Positive ions (cations) keep the name of the atom they are made from. E.g. copper becomes copper ion
Negative ions (anions) use part of the atom's name and add -ide onto the end. E.g. fluorine becomes fluoride ion
We write out their chemical symbols with a little superscript + or - and the number of electrons they lost or gained.

13
Multiple Choice
Where are valence electrons located in an atom?
On the outermost energy level
On the innermost energy level
In the nucleus
14
Multiple Choice
Atoms from group 2 on the Periodic Table are likely to form ions of what charge?
+1
+2
0
−1
−2
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
Fluorine, like all halogens, has 7 valence electrons but wants a full shell. What charge will fluorine make?
7+
7-
1-
1+
18
Extra: forming bonds
to lose or gain electrons, they need to go somewhere
they often go to other atoms who need to lose or gain an electron
this is called chemical bonding, the sharing or exchange of electrons
atoms will balance each other out to form stable valence electron shells

19
Multiple Choice
20
Multiple Choice
Bohr Models + Ion Formation

Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

17 questions
Percent Composition
Presentation
•
9th - 12th Grade

16 questions
Periodic Table Groups
Presentation
•
7th - 12th Grade

19 questions
Structure of Atom
Presentation
•
9th - 12th Grade

14 questions
Acid Naming & Formula Writing
Presentation
•
10th - 11th Grade

14 questions
Animal Farm Ch. 1 Review Quiz
Presentation
•
8th - 10th Grade

13 questions
Periodic Table: Electron Configuration
Presentation
•
8th - 12th Grade

20 questions
Electron Configuration
Presentation
•
9th - 12th Grade

16 questions
U3L3 - Lecture Notes - Electron Configurations
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade