

Chemical Bonding Electron Dot Diagram
Presentation
•
Chemistry
•
9th Grade
•
Hard
Joseph Anderson
FREE Resource
9 Slides • 7 Questions
1
Lewis Electron Dot Structure
Mrs. Perlita V. Sanchez
Grade 9 Science
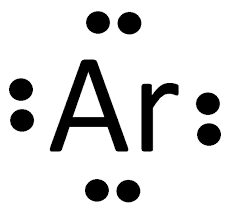
2
I. OBJECTIVES
Illustrate Lewis Electron Dot Structure of some elements.
Understand the Octet Rule
Identify which element or atom will more likely give or accept electrons
3
Lewis Electron Dot Structure
Proposed by Gilbert Lewis
A model used to illustrate the electrons in the outermost energy level of an atom
It helps picture out the Octet Rule and how chemical bonding takes place between elements
4
Lewis Electron Dot Structure
The chemical symbol of an element is surrounded by a number of dots which are the valence electrons.

5
Multiple Choice
The outermost electrons of an atom are __________.
outermost electron
Lewis electrons
valence electrons
octet electrons
6
Octet Rule or Rule of Eight
Elements with with complete number of electron are those with 8 valence electrons and they are described as STABLE ELEMENTS
Example: 18Ar 2) 8) 8) valence electron is 8 therefore 18Ar is stable.
Elements with incomplete number of valence electrons are those with less than 8 valence electrons and they are described as UNSTABLE ELEMENTS
Example: 11Na 2) 8) 1) valence electron is 1, its is less than 8 therefore 11Na is unstable.
7
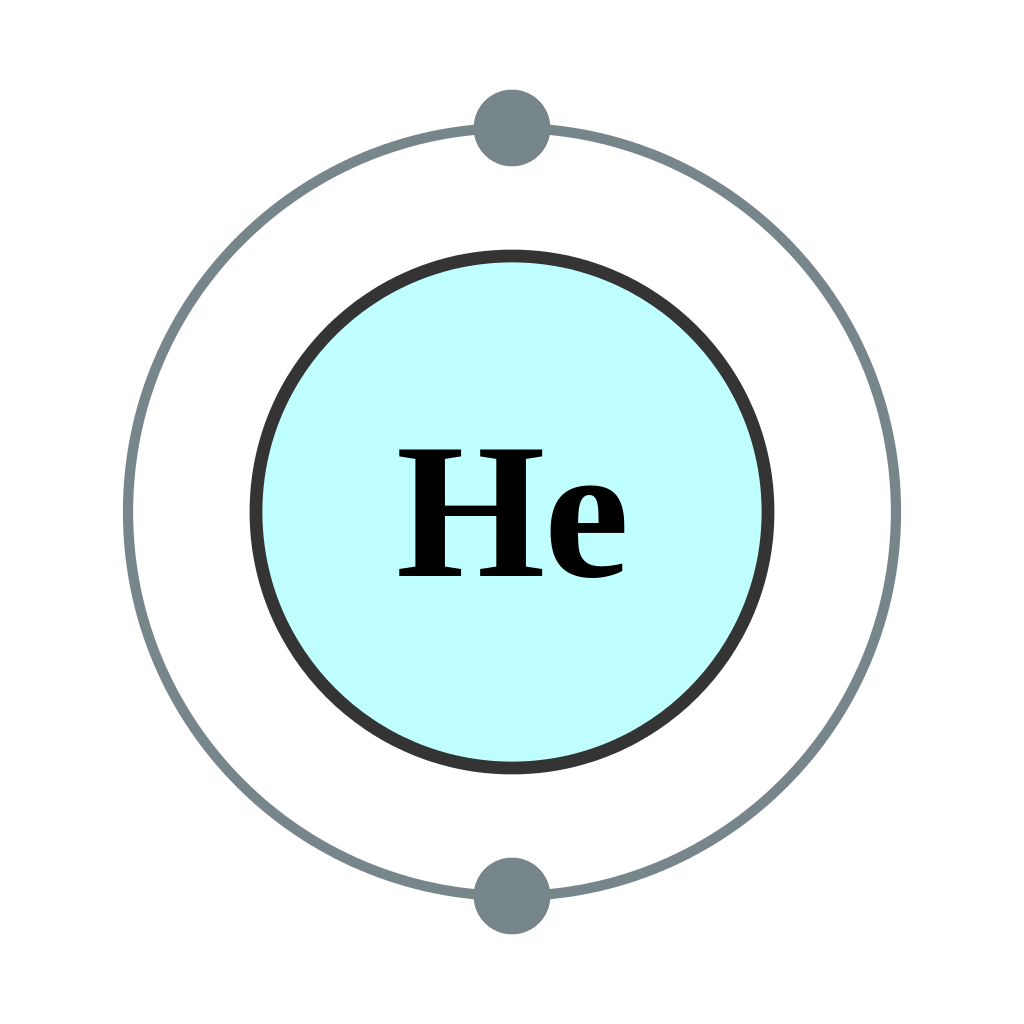
Octet Rule Exemption
The element Helium (2He) is exemption to this rule. Since helium has 2 electrons, its energy level is complete with 2 valence electrons making it a stable element.
8
Multiple Choice
Identify if this element 19K 2) 8) 8) 1) is stable or unstable?
Stable
Unstable
9
Multiple Choice
Identify if the element 10Ne 2) 8) is stable or unstable?
Stable
Unstable
10
Multiple Choice
How many valence electrons should an element have for it to be considered as a STABLE element?
2
10
6
8
11
Accept, Lose or Share?
Elements with fewer electrons tend to lose electrons rather than keeping them because in this way they can achieve a stable condition.
On the other hand, elements with more valence electrons will ten to accept electrons from other elements because they only need fewer electrons to complete their valence electrons.
In the case of carbon atom with 4 valence electrons, it will either lose or accept electron but it will enter into sharing of electrons.
12
Stable Elements
Stable elements DOES NOT accept, lose or share electron because their valence shell is already complete.
13
Multiple Choice
Which of the elements will share electrons?
12Mg 2) 8) 2)
14Si 2) 8) 4)
17Cl 2) 8) 7)
15P 2) 8) 5)
14
Multiple Choice
Which element will most likely lose its electrons?
Na 2)8)1)
N 2)5)
O 2) 6)
F 2)7)
15
Multiple Choice
Which element will prefer to accept electrons from another element?
11Na 2) 8) 1)
20Ca 2) 8) 2)
16S 2) 8) 6)
10Ne 2) 8)
16
Key Concepts
Stability of an element can be identified by knowing how many electrons can be found in its outermost energy level.
The Octet Rule or Rule of Eight, states that for an element to become stable it should have eight valence electrons in its outermost energy level; if not then, it is unstable. With the exemption of Helium.
The Lewis Electron Dot Structure is used to show the valence electron of an element or atom helps us visualize the Octet Rule.
Lewis Electron Dot Structure
Mrs. Perlita V. Sanchez
Grade 9 Science
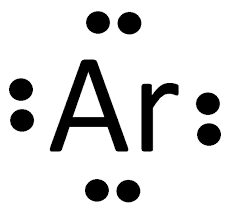
Show answer
Auto Play
Slide 1 / 16
SLIDE
Similar Resources on Wayground

11 questions
Storytelling 9D
Presentation
•
9th Grade

10 questions
culture extension
Presentation
•
9th Grade

12 questions
Atomic Form Protons Neutrons Electrons
Presentation
•
9th Grade

12 questions
Atoms Protons Neutrons Electrons
Presentation
•
9th Grade

15 questions
Exothermic and endothermic reactions
Presentation
•
9th Grade

12 questions
Comma Rules
Presentation
•
10th Grade

11 questions
Crude Oil and Hydrocarbons
Presentation
•
10th Grade

16 questions
Elements, compounds and mixtures
Presentation
•
8th - 10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade

15 questions
Unit 9 solubility curves
Presentation
•
9th Grade - University

39 questions
Nuclear Equations
Quiz
•
9th - 11th Grade

25 questions
Unit 12: Acid-Base Chemistry
Quiz
•
9th - 12th Grade

10 questions
24-25 Nuclear Fusion and Fission Concepts
Interactive video
•
9th - 12th Grade

18 questions
Solutions Chemistry
Quiz
•
9th - 12th Grade

10 questions
Balancing Chemical Equations Challenge
Interactive video
•
6th - 10th Grade