

Electron Cloud
Presentation
•
Chemistry
•
11th Grade
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 6 Questions
1
Chemistry
Electron Configuration
How are electrons arranged in the electron cloud model instead of the Bohr model

2
Bohr vs. Electron Cloud
What is the difference between Bohr's model and the Electron Cloud Model?

3
Niel Bohr's Model (1913)
* Electrons orbit the nucleus in ciruclar paths of fixed energy (energy levels).
* useful for understanding properties of elements and their chemical interactions
*it doesn’t explain certain behaviors of electrons, except for those in the simplest atom, the hydrogen atom.

4
Electron Cloud Model (Erwin Schrödinger-1926)
*The electron cloud model uses the basic idea of Bohr's model except that electrons are not found in distinct orbits but their position can be thought of as in a cloud that has a particular energy.
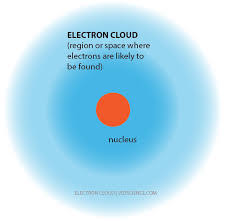
5
Electron Cloud Model
* the nucleus contains protons and neutrons
* a visual model of the probable locations of electrons in an atom
*currently the accepted model of an atom

6
How did Schrödinger proposed electrons travel?
Electrons travel in waves, which means their exact positions cannot be determined.

7
Where would an electron most likely be?
Schrödinger developed an equation that could be used to calculate the chances of an electron being in any given place around the nucleus. Based on his calculations, he identified regions around the nucleus where electrons are most likely to be. He called these regions orbitals.

8
Orbitals
Orbitals may be shaped like spheres, dumbbells, or rings. In each case, the nucleus of the atom is at the center of the orbital.

9
Multiple Choice
In Bohr's model electrons orbit the nucleus in circular paths of _______energy levels.
temporary
fixed
10
Multiple Choice
What is the area surrounding the nucleus of an atom where electron are likely to be?
electron cloud
nucleus
11
Multiple Choice
Which model is the accepted model of an atom?
Bohr Model
Electron Model
12
Multiple Choice
Schrödinger proposed electrons travel in __________.
circular paths
waves
definite paths
13
Multiple Choice
What is the region in the electron cloud around the nucleus of an atom where electrons are most likely to be?
iCloud
orbital
protons
14
Multiple Choice
Orbitals may be shaped like _______, _______, or _______.
squares, dumbbells, rings
spheres, diamonds, rings
spheres, dumbbells, rings
Chemistry
Electron Configuration
How are electrons arranged in the electron cloud model instead of the Bohr model

Show answer
Auto Play
Slide 1 / 14
SLIDE
Similar Resources on Wayground

11 questions
Drawing Bohr Model
Lesson
•
11th - 12th Grade

12 questions
Binary Ionic Formulas and Names
Lesson
•
10th Grade

13 questions
Unraveling the Electron Dance
Lesson
•
11th Grade

15 questions
New Electron Models
Lesson
•
9th - 12th Grade

10 questions
Periodic Table and Elements
Lesson
•
10th - 12th Grade

12 questions
Electron Configuration
Lesson
•
11th - 12th Grade

10 questions
Periodic Trends
Lesson
•
11th - 12th Grade

11 questions
Benzene and Its Derivative
Lesson
•
11th - 12th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

15 questions
Hargrett House Quiz: Community & Service
Quiz
•
5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

15 questions
Equivalent Fractions
Quiz
•
4th Grade
Discover more resources for Chemistry

30 questions
Element compound mixture review quiz
Quiz
•
10th - 12th Grade

20 questions
U4 Reaction Types
Quiz
•
10th - 12th Grade

25 questions
Potential Energy Diagram Checkpoint
Quiz
•
11th Grade

46 questions
Quantum, E Config, Periodic Trends
Quiz
•
11th Grade

5 questions
DOL REC: Solubility Rules and Precipitation Reactions
Quiz
•
10th - 12th Grade

87 questions
Test 3
Quiz
•
9th - 12th Grade

7 questions
Determine Molecular Formula from Empirical
Quiz
•
9th - 12th Grade

15 questions
9-01: Properties of Acids & Bases
Quiz
•
9th - 12th Grade