

Introduction to Radioactivity
Presentation
•
Chemistry
•
10th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
19 Slides • 14 Questions
1
Day 1 - Introduction to Nuclear Radiation

2

3

4
Multiple Choice

What do these isotopes of lithium all have in common?
neutrons & mass number
atomic number and neutrons
atomic number and electrons
protons, atomic number, and mass number
5

6

7
Multiple Choice

The photo above shows the isotopic notation for which isotope?
Carbon 12
Carbon 13
Carbon 14
Carbon 15
8
Multiple Choice
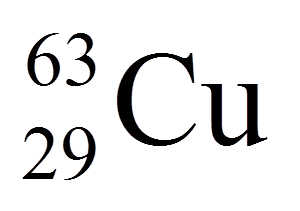
The top number in this isotope notation is
the mass number
the atomic number
the atomic mass
the neutron number
9
Multiple Choice
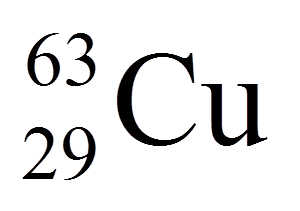
The bottom number in this isotope notation is
the mass number
the atomic number
the atomic mass
the neutron number
10

11

12

13
Multiple Choice
Which of the following is a characteristic of a nuclear reaction?
results from a change in the neutrons or protons of an element
are unaffected by pressure or temperature
cannot be sped up or slowed down
results from changing the mass number of an atom
14

15
Multiple Choice
What is an atom that has an unstable nucleus and undergoes radioactive decay?
Radiation
Radioactivity
Radioisotopes
Radium
16

17

18

19

20
Multiple Choice
X → 86Rn222 + 2He4
21
Multiple Choice
22

23

24
Multiple Choice
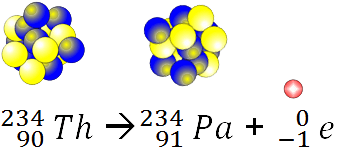
25
Multiple Choice

What was the parent element in this reaction?
23490Th
23892U
23492U
23290Th
26
Multiple Choice

What was the parent element in this reaction?
23490Th
23892U
23492U
23290Th
27
Multiple Choice
Which of the following correctly describes the charge of beta particles and alpha particles?
Alpha particles are positively charged, and beta particles are negatively charged.
Alpha particles and beta particles are both negatively charged.
Alpha particles are negatively charged, and beta particles are positively charged.
Alpha particles and beta particles are both positively charged
28

29

30

31
Fill in the Blanks

Type answer...
32
Multiple Choice
33

Day 1 - Introduction to Nuclear Radiation

Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

22 questions
Accuracy v. Precision, Sig Figs, and Scientific Notation
Presentation
•
9th - 12th Grade

27 questions
Moles - Part 2
Presentation
•
9th - 12th Grade

24 questions
Parallel Lines, Angles, & Transversals
Presentation
•
10th - 12th Grade

24 questions
Direct Object Pronouns
Presentation
•
10th - 12th Grade

25 questions
Electron Configuration, Orbital Notation, & Dot diagrams
Presentation
•
9th - 12th Grade

26 questions
Stoichiometry Lesson
Presentation
•
9th - 12th Grade

27 questions
Bonding and Formulas
Presentation
•
9th - 12th Grade

25 questions
3.4 Dark Reaction / Calvin Cycle
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade