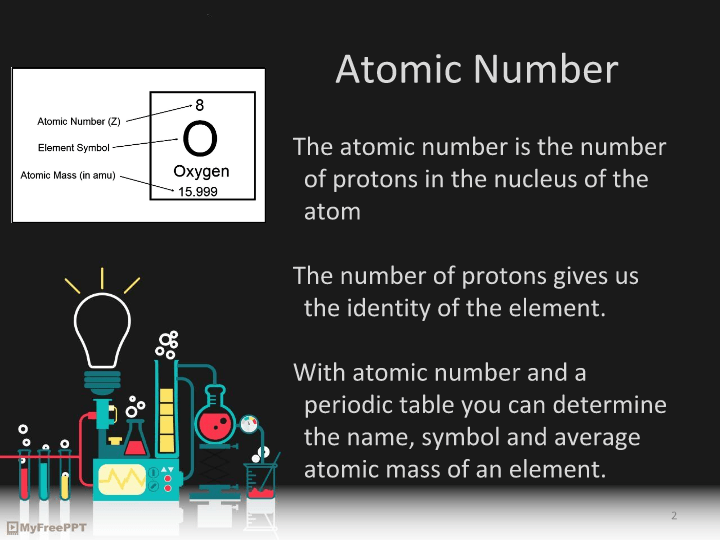
Isotopes, Mass Number, Atomic Number
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
10 Slides • 10 Questions
1
2
3
4
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
10
11
12
13
14
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

16 questions
Air Masses
Lesson
•
9th - 12th Grade

16 questions
Linear and Exponential Functions
Lesson
•
9th - 12th Grade

15 questions
7.4: Percent Composition and Empirical Formula
Lesson
•
9th - 12th Grade

17 questions
Gas Laws - Dalton's Partial Pressure
Lesson
•
9th - 12th Grade

17 questions
Nuclear Decay
Lesson
•
9th - 12th Grade

18 questions
Quantitative vs Qualitative observations
Lesson
•
9th - 12th Grade

14 questions
L1 Types of Matter
Lesson
•
10th - 12th Grade
Popular Resources on Wayground

7 questions
History of Valentine's Day
Interactive video
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

15 questions
Valentine's Day Trivia
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

19 questions
Stoichiometry, Limiting Reactants, and Percent Yield
Quiz
•
10th Grade

20 questions
Stoichiometry Practice
Quiz
•
12th Grade

10 questions
Formative 3BD: Ionic Bonds
Quiz
•
9th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

10 questions
Identifying types of reactions
Quiz
•
9th - 12th Grade