

Kinetic Molecular Theory and the Ideal Gas Laws
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
13 Slides • 27 Questions
1
Gas Laws

2

3
4
Multiple Choice
Which of the following is an assumption of the kinetic-molecular theory of gases?
Collisions between gas particles are inelastic.
Gases consist of closely spaced particles.
Gas particles move around in an orderly manner.
The temperature of a gas depends on the average kinetic energy of the gas particles.
5
Multiple Choice

These properties describe a ___.
solid
liquid
gas
6
Multiple Choice

These properties describe a ___.
solid
liquid
gas
7
Multiple Choice

These properties describe a ___.
solid
liquid
gas
8

9

10
11

12
Multiple Choice
How many mm Hg is a pressure of 1.05 atm?
529 mm Hg
798 mm Hg
104.8 mm Hg
760 mm Hg
13
Multiple Choice
What does STP (standard temperature and pressure) stand for?
0.0 degree Celcius and 1 atm
1.0 mmHg and 273 degree Kelvin
1.0 degree Celcius and 0 atm
1.0 atm 273 degree Celcius
14
Multiple Choice
A gas is at a pressure of 95 kPa. What is its pressure in atmospheres?
1.5 atm
1.09 atm
0.94 atm
0.82 atm
15
Multiple Choice
760 mmHg is equal to
1 torr
760 atm
760 torr
101 Pa
16
Multiple Choice
Which of the follow is NOT equal to 1 atm?
14.7 psi
760 mmHg
101325 Pa
101.325 torr
17
Multiple Choice
Solids are described as having a
definite shape and indefinite volume
indefinite shape and indefinite volume
definite shape and definite volume
indefinite shape and definite volume
18
Multiple Choice
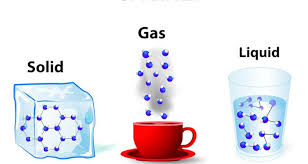
Of these three states of matter, which one has the most kinetic energy?
Solid
Liquid
Gas
19
Multiple Select
The properties of liquids include (SELECT ALL THAT APPLY)
definite volume
take the shape of the container
can flow
are relatively incompressible
can diffuse and have surfacre tension
20
Multiple Select
According to the Kinetic Molecular Theory a liquid's (SELECT ALL THAT APPLY)
particles are held close together by intermolecular forces
particles are in constant motion
particles are vibrating in place and closely packed
particles are not moving
21
Multiple Choice

Flask 1 contain the most molecules
Flask 2 contain the most molecules
Flask 3 contain the most molecules
Flask 4 contain the most molecules
22
Multiple Choice
Gas pressure is caused by
gas molecules colliding with surfaces
gas molecules condensing to a liquid
gas molecules hitting other gas molecule
barometers
23

24

25

26

27
28
29
Multiple Choice
Convert: -45.9 C to K:
227.1 K
400 K
0 K
373 K
30
Multiple Choice
Convert: 186.5 K to C
-86.4 C
-50 C
-68.4 C
180 K
31
Multiple Choice
Convert: 0 K to C
-273 C
-273 K
273 C
273 K
32
Multiple Choice
Convert: 253 C to K:
526 K
625 K
0 K
186 K
33
Multiple Choice

Select the temperature in °C
50°C
10°C
20°C
60°C
34
Multiple Choice

Select the temperature in °C
30°C
100°C
90°C
40°C
35
Multiple Choice

The faster molecules move,
the higher the temperature
the less temperature
the same temperature
I don't know
36
Multiple Choice

The absolute coldest anything can get.
In Celsius it's at 273 degrees. Or 0 degrees Kelvin.
Absolute cold
Kelvin
Absolute zero
I don't know
37
Multiple Choice

Scientists' favorite temperature scale, because it begins at the absolute coldest anything can get.
Celsius
Fahrenheit
Kelvin
All of them
38
Multiple Choice

Which direction is heat flowing in the picture?
from the hand to the ice
from the ice to the hand
heat is not being transferred
there is no way to tell
39
Multiple Choice
The measure of average kinetic energy of all the particles within an object is called ____________.
temperature
conduction
radiation
heat
40
Multiple Choice
In nature, heat always flows from a:
cold object to a warm object.
small object to a big object.
warm object to a cold object.
large object to a small object.
Gas Laws

Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

35 questions
Bonding (Covalent and Ionic Bonds)
Presentation
•
9th - 12th Grade

36 questions
Periodic Table Trends. Cole
Presentation
•
10th Grade - University

34 questions
Ionic and Covalent Bonding Review Lesson
Presentation
•
9th - 12th Grade

36 questions
2nd 6wks
Presentation
•
9th - 12th Grade

33 questions
Bonding Lesson Part 2
Presentation
•
9th - 12th Grade

40 questions
Balancing and 5 Types of Chemical Reactions
Presentation
•
9th - 12th Grade

30 questions
Endothermic/Exothermic & Calorimetry
Presentation
•
9th - 12th Grade

37 questions
ServSafe - Food Handler 4b - TCS Foods
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
Balancing and Reaction Types
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

16 questions
STAAR Review Quizziz 5
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

5 questions
DOL REC: pH and pOH Calculations
Quiz
•
10th - 11th Grade

11 questions
Balancing Chemical Equations
Presentation
•
9th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

5 questions
DOL PreAP: pH and pOH Calculations
Quiz
•
10th Grade