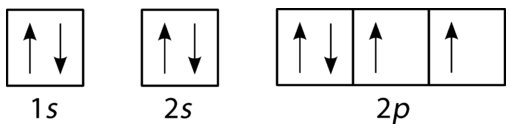
Principle Energy Level
Presentation
•
Chemistry
•
11th Grade
•
Hard
Joseph Anderson
FREE Resource
15 Slides • 18 Questions
1
2
3
4
5
6
Multiple Choice
7
8
Fill in the Blanks
9
Fill in the Blanks
10
Fill in the Blanks
11
12
13
14
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
19
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
24
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
30
Multiple Choice
31
Multiple Choice
32
Multiple Choice
33
Multiple Choice
Show answer
Auto Play
Slide 1 / 33
SLIDE