

Rates of Reactions
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 13 Questions
1
Reaction rates
2

3

4

5
Multiple Choice
true
false
6
Multiple Choice
The energy needed for a reaction to occur
The minimum amount of energy needed for a reaction to potentially occur
The energy added by a catalyst
The energy possessed by the products
7
Multiple Choice
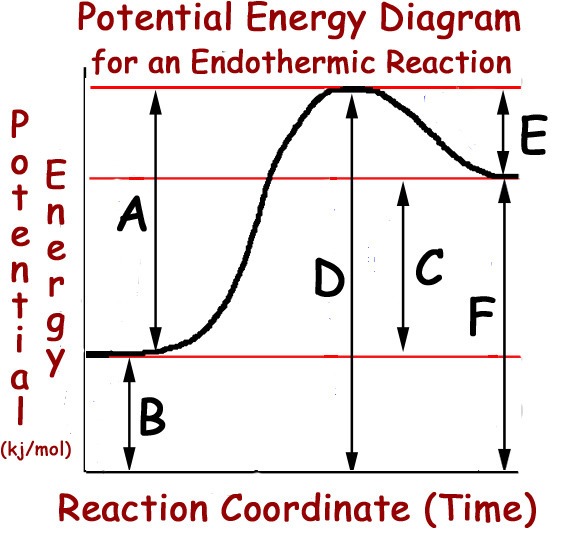
A
B
C
D
8
Multiple Choice
What is collision theory?
Molecules must collide in the correct orientation with enough energy to bond.
Molecules need enough energy to collide and react.
Atoms constantly collide and react.
The minimum energy needed for atoms to react
9
Multiple Choice
True
False
10
Multiple Choice
What is the rate of reaction?
How much energy is needed for a reaction to occur.
The energy required to break a bond.
The time it takes for a reaction to occur.
Collision Theory
11
12

13

14

15
Multiple Choice
What is the purpose of a catalyst?
Helps to slow down a reaction
Raises the activation energy
Lowers the activation energy
Is consumed by the reaction
16
Multiple Choice
What factors effect rate of reaction?
Temperature, Concentration, Pressure and Energy
Surface Area, Concentration, Energy and Pressure
Temperature, Pressure, Concentration and Surface area
Pressure, Surface area, Density and Energy
17
Multiple Choice
Increasing the temperature of your solution will.......
Not affect the rate of reaction.
Speed up the rate of reaction
Slow down the rate of reaction
18
Multiple Choice
Slow collision frequency
Allow less effective collision between the particles
Cause particles to lose speed
Increase collision between the particles thus increasing the rate.
19
Multiple Choice
concentration
surface area
temperature
reactants
20
Multiple Choice
catalyst
product
reactant
solute
21
Multiple Choice
it increases the amount of reactants
it lowers the activation energy
it increases the energy of particle collisions
it increases the frequency of particle collisions because there are more collision sites
Reaction rates
Show answer
Auto Play
Slide 1 / 21
SLIDE
Similar Resources on Wayground

16 questions
La Francophonie
Presentation
•
9th - 12th Grade

16 questions
Indefinite and negative words in Spanish
Presentation
•
9th - 12th Grade

17 questions
Percent Composition
Presentation
•
9th - 12th Grade

19 questions
Structure of Atom
Presentation
•
9th - 12th Grade

16 questions
Mole Conversions & Percent Composition Practice
Presentation
•
10th - 12th Grade

20 questions
Electron Configuration
Presentation
•
9th - 12th Grade

14 questions
Acid Naming & Formula Writing
Presentation
•
10th - 11th Grade

16 questions
British Values, Prevent and Radicalisation Review
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

120 questions
2nd Semester Final Review
Quiz
•
11th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade