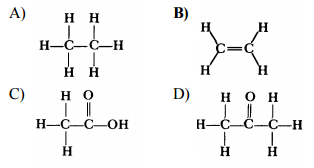
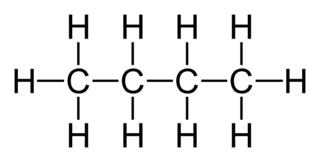
Substituting Hydrocarbons
Presentation
•
Chemistry
•
10th - 11th Grade
•
Hard
Joseph Anderson
FREE Resource
12 Slides • 15 Questions
1
2
3
Poll
4
5
Multiple Choice
6
Multiple Choice
7
8
9
10
11
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
24
25
26
Poll
27
Show answer
Auto Play
Slide 1 / 27
SLIDE
Similar Resources on Wayground

20 questions
Periodic Families
Lesson
•
9th - 10th Grade

25 questions
Covalent Bonds
Lesson
•
10th - 11th Grade

20 questions
Factoring Polynomials: Special Cases
Lesson
•
10th - 12th Grade

20 questions
Work and Power
Lesson
•
10th - 12th Grade

22 questions
Thermochemistry & Nuclear Chemistry Review
Lesson
•
10th Grade

21 questions
Cold War Intro
Lesson
•
KG

21 questions
Triangle Sum/Exterior Angle Theorem
Lesson
•
10th - 11th Grade

23 questions
Chemical Equilibrium 13.1-13.3 Timberlake
Lesson
•
10th - 11th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

20 questions
Chemistry: Classification of Matter
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade

20 questions
Stoichiometry Review
Quiz
•
10th Grade