

Covalent Bonds
Presentation
•
Chemistry
•
10th - 11th Grade
•
Practice Problem
•
Medium
+3
Standards-aligned
Stacy King
Used 46+ times
FREE Resource
15 Slides • 10 Questions
1
Covalent Bonds

2
Covalent Bond
formed between nonmetals

3
Covalent Bond
electronegativity attraction forces being stronger than the repulsion forces

4
Covalent Bond
Low melting point
do not conduct electricity
primarily gases
5
Covalent bond
atoms with high kinetic energy react with other atoms to become more stable

6
Covalent Bond
bond energy is the amount of energy to break a chemical bond

7
Covalent Bond
Electron Dot or Lewis Dot
signifies the bonding pair of electrons

8
covalent bond
polar- unequal sharing of electrons
creates dipoles

9
Covalent bond
non polar is equal sharing of electrons

10
Covalent Bond
Electron Dot or Lewis Dot
signifies the bonding pair of electrons

11
Polyatomic ions
a bonded set of 2 or more atoms that function as a single unit

12
Covalent Bonds
Resonance/hybrid Structures
are electron dot structures that have multiple correct lewis dot arrangements

13
Octet Exceptions
Free Radicals have odd number of electrons

14
Octet exceptions
electron deficient the central atom does not have a filled valence shell

15
Octet Exceptions
hypervalency atoms can have more than 8 electrons

16
Multiple Choice
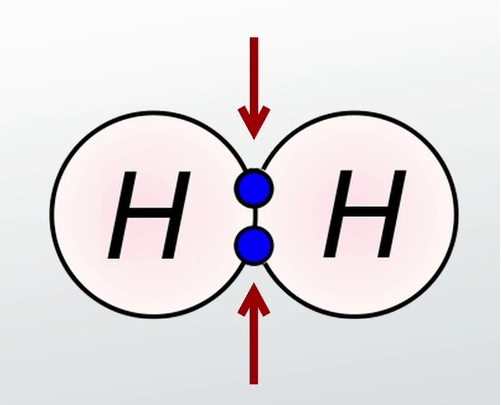
17
Multiple Choice

18
Multiple Choice
19
Multiple Choice
20
Multiple Choice

21
Multiple Choice

22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
Valence electrons are found
in the innermost energy level of an atom
in the middle energy levels of an atom
in the outermost energy levels of an atom
25
Multiple Choice
Covalent Bonds

Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

20 questions
Water, Solubility, Solutions, Molarity
Presentation
•
9th - 12th Grade

22 questions
Classifying Reactions
Presentation
•
10th Grade

17 questions
Mass and Percent Composition
Presentation
•
10th Grade

20 questions
Oxidation State Practice
Presentation
•
10th - 12th Grade

20 questions
Covalent Bonds Lesson
Presentation
•
10th Grade

20 questions
DLD Performance Final
Presentation
•
9th - 12th Grade

17 questions
Lesson: Ions POGIL
Presentation
•
10th - 12th Grade

20 questions
Atomic Structure Review
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade