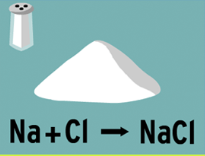
TMSCA Science Lesson
Presentation
•
Science
•
7th - 9th Grade
•
Hard
Joseph Anderson
FREE Resource
5 Slides • 40 Questions
1
2
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice
37
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
Show answer
Auto Play
Slide 1 / 45
SLIDE
Similar Resources on Wayground

41 questions
Characteristics of Living Things
Lesson
•
6th - 8th Grade

38 questions
Force and Motion
Lesson
•
8th Grade

38 questions
Electricity Lesson
Lesson
•
6th - 8th Grade

36 questions
Natural Selection Info
Lesson
•
7th - 8th Grade

40 questions
Sci 8 1.2 Scientific Method
Lesson
•
8th Grade

43 questions
Change over Time
Lesson
•
8th Grade

39 questions
Waves
Lesson
•
6th - 8th Grade

39 questions
Atomic Structure
Lesson
•
8th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Science

22 questions
Phases of the moon
Quiz
•
8th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

20 questions
Flow of Energy
Quiz
•
7th Grade

12 questions
Ecological Succession
Quiz
•
7th Grade

20 questions
Waves and Wave Properties
Quiz
•
6th - 8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

10 questions
Understand Ecosystem Roles and Energy Flow
Interactive video
•
6th - 8th Grade

10 questions
Exploring the Rock Cycle: Types and Formation
Interactive video
•
6th - 8th Grade