

Unit 8: Electrons and the Periodic Table
Presentation
•
Chemistry
•
9th - 12th Grade
•
Easy
+5
Standards-aligned
Meredith Bizragane
Used 2+ times
FREE Resource
8 Slides • 67 Questions
1
Atomic Structure
By Meredith Bizragane
Nucleus of an atom is central and dense and holds the protons and neutrons.
Atomic number = number of protons
Atomic mass (mass number) = protons + neutrons
Neutral atoms: # of Protons = # of electrons
Isotopes: Atoms of the same elements with a different atomic mass.
2
Multiple Choice
What is the atomic number?
the number of protons in the nucleus
the number of protons and neutrons in the nucleus
the number of neutrons in the nucleus
the number of protons in the energy levels
3
Multiple Choice
What is the mass number (atomic mass)?
the number of protons in the nucleus
the number of protons and neutrons in the nucleus
the number of neutrons in the nucleus
the number of protons and electrons in the atom
4
Multiple Choice
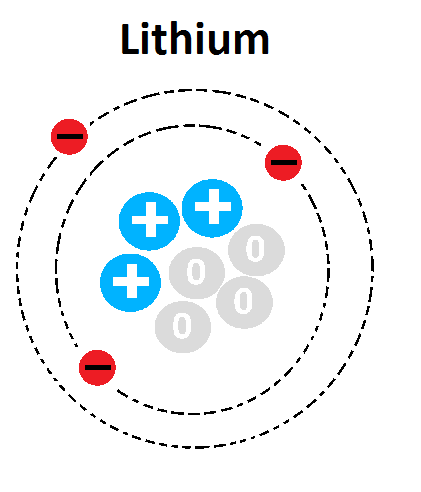
What is the atomic number of this atom?
1
3
4
7
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
An atom has 10 protons, 15 neutrons and 10 electrons what is its mass number.
20
10
35
25
10
Multiple Choice
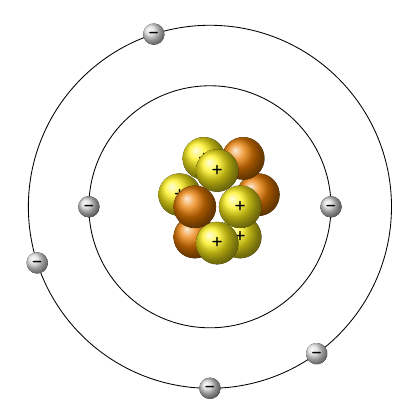
How many electrons does this atom have?
2
4
6
10
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice

14
Multiple Choice
Isotopes of an element have a different number of...
Protons
Neutrons
Electrons
Mass
15
Multiple Choice
Potassium-39 has how many neutrons?
19
18
20
21
16
Multiple Choice
Identify the element and its atomic mass:
76 protons and 114 neutrons
Osmium-114
Osmium-76
Osmium-190
Osmium-190.23
17
Multiple Choice
An atom of an element with a different number of neutrons and therefore a different atomic mass
Isotope
Ion
Atom
Particle
18
Multiple Select
Which of the following have the same MASS NUMBERS? (You must choose more than one answer)
scandium - 46
sodium - 21
titanium -46
boron - 11
19
Multiple Choice

One atom has 56 protons and 18 neutrons.
A different atom has 56 protons and 17 neutrons. These aroms are:
different elements
isotopes
20
Electrons
Bohr Model: electrons orbit the nucleus in energy levels
Quantum Mechanical Model: electrons are in s, p, d, and f orbitals
21
Multiple Choice

1s22s22p63s23p64s23d10
22
Multiple Choice
Each row on the periodic table represents:
an energy level
a sublevel
an electron
an orbital
23
Multiple Choice
24
Multiple Choice

25
Multiple Choice
26
Multiple Choice
1s22s22p63s2
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice
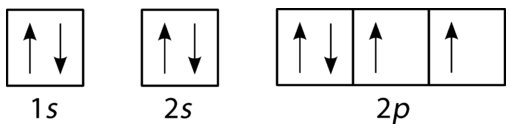
30
Multiple Choice

31
Multiple Choice

32
Multiple Choice

Which example shows a violation of Hund's Rule?
A
B
C
D
33
Multiple Choice

Which of the following is written correctly?
A
B
C
D
34
Multiple Choice

What element's orbital notation is pictured here?
Ar
Br
Cu
Cl
35
Multiple Choice

Which element is this?
Nickel
Neon
Sodium
Nitrogen
36
Multiple Choice

This could be the dot diagram of
Mg
Cl
C
O
37
Multiple Choice

This is a correct dot diagram for neon (Ne)
true
false
38
Multiple Choice
How would you draw a Lewis dot diagram for Magnesium?
39
Multiple Choice
Which electron configuration belongs to a Chloride (Cl-) ion?
1s2 2s2 2p6 3s2 3p5
1s2 2s2 2p6 3s2 3p6
1s2 2s2 2p6 3s2 3p7
1s2 2s2 2p6 3p7
40
Multiple Choice
41
Multiple Choice

42
Periodic Trends
Atomic radius decreases across a row since the electrons are on the same energy level but the effective nuclear charge increases as protons are added to the nucleus. This increasing the force of attraction and pulls electrons closer to the nucleus.
Atomic radius increases down a column since electrons are added to higher energy levels therefore increasing the distance from the nucleus to the valence electrons. Even though elements farther down the periodic table have more protons the distance outweighs the charge.
43
Periodic Trends
First ionization energy increases as atomic radius decreases.
It is harder to remove an electron from a smaller atom since more energy is required to overcome the stronger force of attraction between electrons and protons in smaller atoms
44
Multiple Choice
What is the relationship between effective nuclear charge and ionization energy?
The ionization energy decreases with an increase in effective nuclear charge.
There is no relationship between effective nuclear charge and ionization energy.
Effective nuclear charge and ionization energy are unrelated.
The ionization energy increases with an increase in effective nuclear charge.
45
Multiple Choice
Which element has the highest effective nuclear charge: carbon, nitrogen, or oxygen?
oxygen
hydrogen
nitrogen
carbon
46
Multiple Choice
Recap:
Coulomb’s law states that the force between two charged objects will __________ when the magnitude of the object’s charge increases.
Coulomb’s law also states that the force between two charged objects will __________ when the distance between objects increases.
increase; increase
increase; decrease
decrease; increase
decrease; decrease
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Electromagnetic Radiation
c=λν E=hν
Wavelength (λ): distance between crests of adjacent waves measured in units of meters or nanometers.
Frequency (ν): the number of waves that pass a point per second measured in units of Hz or /s
Energy (E): Plank's constant times frequency
Speed of light (c): 3.00x108m/s
Plank's Constant (h): 6.626x10-34 Js
1 m = 1x109 nm
52
Electromagnetic Radiation
Wavelength is inversely proportional to energy and frequency.
Energy and frequency are directly proportional.
Visible light is 400 nm (violet) to 700 nm (red).

53
Electromagnetic Radiation
When atoms absorb energy, electrons jump to excited states in absorption. This energy is then released as the electron returns to a lower energy state and finally the ground state during emission.
The longer the distance the electron travels, the more energy is involved in the transition.
Higher energies correspond to lower wavelengths.

54
Electromagnetic Radiation
Absorption and emission data can be collected and shown on a spectrum. Absorption spectrum show black lines on a colored background and emission spectrum show colored lines on a black background.
Each spectral line corresponds to a specific energy transition.
Since each element has a unique spectrum, element's can be identified in this way.

55
Multiple Choice
Calculate the wavelength of the yellow light emitted by the street light, if the frequency of the radiation is 5.10x1014Hz.
5.12 x 10-7m
5.88 x 10-7 m
4.20 x 1014m
3.0 x 108m
56
Multiple Choice
Energy is measured in........
Wavelength
Meters
Joules
Frequency
57
Multiple Choice
The symbol for wavelength Is?
nu
lambda
vi
meters
58
Multiple Choice
On a wave, the distance between crest to crest is called?
amplitude
wavelength
frequency
trough
59
Multiple Choice
Which color in the visible spectrum of light has the longest wavelength?
Indigo
Red
Blue
Violet
60
Multiple Choice
Calculate the wavelength of blue light emitted by a mercury lamp with a frequency of 6.88 x 1014 Hz.
3.44 x 10-6m
4.36 x 10-7m
3.0 x 108 m
633 m
61
Multiple Choice
What is the frequency of light with a wavelength of
4.8 x 10-7 meters?
5.34 x 10-9 Hz
3.0 x 108 Hz
3.21 x 1017 Hz
6.25 x 1014 Hz
62
Multiple Choice
The unit of measure for frequency is?
Seconds
Meters
Hertz
Liters
63
Multiple Choice
The symbol for a frequency of a wave is?
Hertz
v
lambda
meters/second
64
Multiple Choice
What does "h" stand for?
Speed of Light
Plank's Constant
Speed of Sound
Constant Energy
65
Multiple Choice
What is the units for speed of light?
c
J
m/s
Hz
66
Multiple Choice
What does energy have to do with wavelength and frequency?
The higher the frequency the less energy the wave has.
The lower the frequency the more energy the wave has.
The shorter the wavelength the more energy the wave has.
The longer the wavelength the more energy the wave has.
67
Multiple Choice

An astronomer looks at the spectrum of light from a distant star. The spectrum they see is shown in the figure. They compare the emission lines within the spectrum to the emission lines in the spectra of pure elements, which are also shown in the figure, in order to identify which elements are present in the outer layers of the star. State all of the elements that are present in the outer layers of the star.
hydrogen, helium, and carbon
hydrogen, helium, and boron
helium, oxygen , and carbon
hydrogen, helium, boron, and carbon
68
Multiple Choice

The absorption lines in the spectrum for Galaxy B are shifted toward the _____ end of the spectrum when compared to the lines for the Milky Way.
red
blue
69
Multiple Choice

70
Multiple Choice

The ground state is the ______________ energy state of an atom.
highest
lowest
71
Multiple Choice

72
Multiple Choice

73
Multiple Choice

74
Multiple Choice

If an electron moves from n=4 to n=2 it ____
absorbs energy
releases energy
75
Multiple Choice

The emission spectrum of carbon is shown. Which spectral lines is associated with an electron making the largest energy jump?
The blue line at 478 nm.
The red line at 658 nm.
The yellow line at 602 nm.
This cannot be determined from an emission spectrum.
Atomic Structure
By Meredith Bizragane
Nucleus of an atom is central and dense and holds the protons and neutrons.
Atomic number = number of protons
Atomic mass (mass number) = protons + neutrons
Neutral atoms: # of Protons = # of electrons
Isotopes: Atoms of the same elements with a different atomic mass.
Show answer
Auto Play
Slide 1 / 75
SLIDE
Similar Resources on Wayground

70 questions
Verbo GUSTAR
Presentation
•
9th - 12th Grade

75 questions
Types, Sources, and History of Law
Presentation
•
9th - 12th Grade

69 questions
10.2 PAST TIME
Presentation
•
9th - 12th Grade

71 questions
Electrostatics
Presentation
•
9th - 11th Grade

70 questions
2.2 Market research
Presentation
•
9th - 12th Grade

72 questions
Ch. 6 The Periodic Table and Periodic Law
Presentation
•
9th - 12th Grade

69 questions
Color Scheme
Presentation
•
9th - 12th Grade

71 questions
Realidades 2 Ch 1B VOCABULARY (Textbook Page 68)
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
Balancing and Reaction Types
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

16 questions
STAAR Review Quizziz 5
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

5 questions
DOL REC: pH and pOH Calculations
Quiz
•
10th - 11th Grade

11 questions
Balancing Chemical Equations
Presentation
•
9th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

5 questions
DOL PreAP: pH and pOH Calculations
Quiz
•
10th Grade