

chem review
Presentation
•
Biology
•
12th Grade
•
Medium
+13
Standards-aligned
Peter Pappagianopoulos
Used 4+ times
FREE Resource
33 Slides • 76 Questions
1

2
Multiple Choice
_____ is the study of the composition, structure, and properties of matter.
Science
Chemistry
Biology
Knowledge
3

4
Multiple Choice
Why is it important to study chemistry in A&P class?
It helps understand body functions
It is not relevant to biology
It focuses only on chemical reactions
It is only about laboratory techniques
5
Multiple Choice
6

7

8
Multiple Choice
9
Multiple Choice

10
Multiple Choice
What is the charge of a proton
positive
negative
neutral
11
Multiple Choice

12
Multiple Choice
13
Multiple Choice

14
Multiple Choice
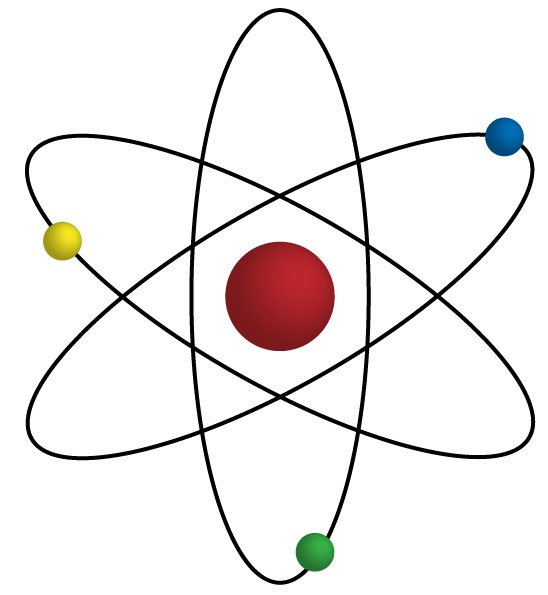
15

16
Multiple Choice
What is the atomic number of an element?
The number of protons in an atom's nucleus
The number of neutrons in an atom's nucleus
The total number of protons and neutrons in an atom
The number of electrons in an atom's outer shell
17
Multiple Choice
If an element has an atomic number of 11, how many protons does it have?
11
12
10
9
18
Multiple Choice

The number 16 for the atomic element sulfur is the
ionic number
isotope
atomic mass
atomic number
19
Multiple Choice

20
Multiple Choice

21

22
Multiple Choice
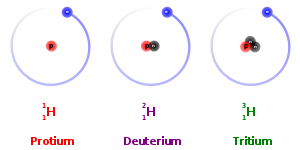
23
Multiple Choice
How many neutrons are in an atom of carbon-14?
6
8
10
14
24
Multiple Choice

25

26

27
Multiple Choice
What are the maximum number of electrons that can be held in the first, second, and third electron shells?
2, 8, 8
2, 4, 6
8, 8, 18
2, 6, 8
28
Multiple Choice

29
Multiple Choice

30

31

32
Multiple Choice
What do atoms do to become stable?
They lose electrons
They take electrons
They share electrons
All of the above
33

34
Multiple Choice

35
Multiple Choice
What is a positive ion called?
anion
cation
isotope
covalent
36
Multiple Select
What are the definitions of electrolytes, acids, bases, and salts as described in the image?
Electrolytes are substances that release ions in water.
Acids are electrolytes that dissociate to release hydrogen ions in water.
Bases are substances that release ions that can combine with hydrogen ions.
Salts are electrolytes formed by the reaction between an acid and a base.
37
Multiple Choice
What is a negative ion called?
anion
cation
covalent
isotope
38
Multiple Choice
How many electrons would a Nitrogen ion gain/lose? If it has 5 valence electrons.
lose 5
gain 5
lose 3
gain 3
39
Multiple Choice
How many electrons would a Calcium ion gain/lose? If it has 2 valence electrons.
lose 1
gain 1
lose 2
gain 2
40
Multiple Choice
Iodine has 7 valence electron. It will have a charge of _______
-2
-1
+1
+2
41
Multiple Choice
Oxygen has 6 valence electron. It will have a charge of _______.
+1
-1
+2
-2
42
Multiple Choice
43

44
Multiple Choice
45
Multiple Choice
46
Multiple Choice

This image shows the bonding between Lithium and Fluorine. What does the red arrow show?
electrons being shared
electrons being transferred to Fluorine
electrons being transferred to Lithium
electrons being destroyed
47
Multiple Choice
48

49
Multiple Choice
How do covalent bonds form?
Donating & receiving valence electrons between atoms
Oppositely charged ions attract each other & form bonds
Scientists are still not sure how they form
Sharing valence electrons between atoms
50
Multiple Choice

This is the displayed formula for what covalent compound?
Water
Hydrogen peroxide
Hydrogen oxide
Ethanol
51
Multiple Choice

Chlorine is a diatomic element which means in nature, it is found in two's. (Cl2) Chlorine shares one pair of electrons to make sure each atom complete the octet rule. This creates a...
single bond
double bond
triple bond
52
Multiple Choice

Oxygen is a diatomic element which means in nature, it is found in two's. (O2) Oxygen shares two pairs of electrons to make sure each atom completes the octet rule. This creates a...
single bond
double bond
triple bond
53
Multiple Choice

Nitrogen is a diatomic element which means in nature, it is found in two's. (N2) Nitrogen shares three pairs of electrons to make sure each atom completes the octet rule. This creates a...
single bond
double bond
triple bond
54
Multiple Select

What does this picture illustrate? (Select all that apply)
Sharing valence electrons
Covalent bonding
transferring valence electrons
ionic bonding
two nonmetals
55
Multiple Choice

How many covalent bonds are created in a single water molecule?
0
1
2
3
4
56

57
Multiple Choice

58

59
Multiple Choice
The electrons in a polar covalent molecule are shared...
Evenly
Unevenly
Electrons are not shared
None of the Above
60
Multiple Choice
The electrons in a nonpolar covalent molecule are shared...
Evenly
Unevenly
Electrons are not shared
None of the Above
61
Multiple Choice
The electrons in an ionic molecule are shared...
Evenly
Unevenly
Electrons are not shared
None of the Above
62
Multiple Choice
In a polar covalent bond, the electrons gather around...
The atom with the Greatest Electronegativity
The atom with the Lowest Electronegativity
Each atom Equally
None of the Above
63
Multiple Choice
The polarity of a bond is determined by...
The sum of the electronegativities of the two atoms
The difference in the electronegativities of the two atoms
The charges of the atoms
None of the Above
64
Multiple Choice
Partial charges are present in which type of bond?
Ionic
Nonpolar Covalent
Polar Covalent
Both Polar Covalent and Ionic
65
Reorder
Use your electronegativity table to rank the following molecules from LEAST to GREATEST polarity.
O2
CCl4
H2O
HCl
BF3
66

67
Multiple Choice
What is a hydrogen bond?
A strong attraction between molecules
A weak attraction between polar molecules
A type of covalent bond
A bond formed only in gases
68

69
Multiple Choice

70

71
Open Ended

Explain what is happening in the following image.
72

73

74
Reorder
Rank the following pH values in order from least (#5) hydronium ion concentration to the greatest (#1)
11
8
6
4
2
75
Multiple Choice
Dehydration synthesis is the reaction that occurs, allowing amino acids to bond. What happens during this process?
add water to build a bond
add water to break a bond
remove water to build a bond
remove water to break a bond
76

77
Multiple Choice
What are the main differences between organic and inorganic molecules?
Organic molecules contain carbon and hydrogen, while inorganic molecules do not.
Organic molecules are generally smaller than inorganic molecules.
Organic molecules do not dissolve in water, while inorganic molecules do.
Organic molecules are only carbohydrates, while inorganic molecules include water and salts.
78

79
Multiple Choice
What is the role of water in living organisms?
It is the most abundant compound
It helps in metabolic reactions
It transports chemicals
All of the above
80

81
Multiple Choice
What are the roles of inorganic salts in the body?
They are waste products
They provide necessary ions
They are not important
They are only found in food
82

83
Multiple Choice
What are the main functions of carbohydrates in living organisms?
Provide energy to cells
Supply materials to build cell structures
Water-insoluble
Contain only carbon
84

85
Multiple Choice
What are the three types of carbohydrates represented in the images?
Monosaccharide
Disaccharide
Polysaccharide
All of the above
86

87
Multiple Choice
What are the building blocks of triglycerides?
1 glycerol and 3 fatty acids
2 glycerol and 2 fatty acids
1 glycerol and 2 fatty acids
3 glycerol and 1 fatty acid
88

89
Multiple Choice

This image is an example of a phospholipid. Which part of a phospholipid is HYDROPHOBIC?
the phosphate "head"
the lipid "tails"
90
Multiple Choice

This image is an example of a phospholipid. Which part of a phospholipid is HYDROPHILIC?
the phosphate "head"
the lipid "tails"
91

92

93
Multiple Choice
Proteins are made of many ____ bonded together.
amino acids
enzymes
polypeptides
94
Multiple Choice
All amino acids have the same basic structure. Which part of the amino acid makes each each one unique?
amino group
carboxyl group
R-group (side chain)
95
Multiple Choice
The protein structure that results from a sequence of amino acids is:
primary structure
secondary structure
tertiary structure
quaternary structure
96

97
Multiple Choice
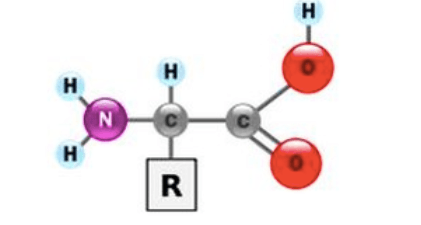
98
Multiple Choice
The protein structure that results from hydrogen bonds forming beta pleated sheets and alpha helices is:
primary structure
secondary structure
tertiary structure
quaternary structure
99
Multiple Choice
What does an enzyme do?
slows down chemical reactions
speeds up chemical reactions
increases the temperature of the reaction
100

101

102
Multiple Choice
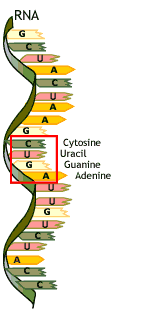
103
Multiple Choice

Nucleic acids
Proteins
Carbohydrates
Lipids
DNA and RNA
104
Multiple Choice
105
Multiple Choice
106
Multiple Choice
107
Multiple Choice
108
Multiple Choice

109


Show answer
Auto Play
Slide 1 / 109
SLIDE
Similar Resources on Wayground

102 questions
Cuối kì I - Khối 12
Presentation
•
12th Grade

104 questions
Apartheid
Presentation
•
KG - University

103 questions
Asian Culture
Presentation
•
KG - University

109 questions
unidad 8 lesson1 and 2 2021-1
Presentation
•
University

108 questions
NE Chat Training
Presentation
•
KG

101 questions
Culture, Race, and Gender
Presentation
•
KG - University

104 questions
Network + Module 1
Presentation
•
University

102 questions
5.5 Operations with Radical Expressions
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Biology

50 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

17 questions
Communicable and Chronic Diseases Slidedeck
Presentation
•
9th - 12th Grade

20 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

64 questions
Disease Unit- 2026
Quiz
•
9th - 12th Grade

55 questions
Mock EOC/ Interim 3 Review
Quiz
•
KG - University

44 questions
Biology EOC sem 2 review
Quiz
•
9th - 12th Grade

45 questions
Ecology Review
Quiz
•
9th - 12th Grade

131 questions
2026 Biology Final Review
Quiz
•
9th - 12th Grade