

CP2 review
Presentation
•
Science
•
6th - 8th Grade
•
Hard
+16
Standards-aligned
Robertha Carmona
FREE Resource
39 Slides • 76 Questions
1
Checkpoint 2 Review
8th Grade Science

2
Counting Atoms - Conservation of Mass

3
How to count atoms in a chemical formaula.
You already know how to read SUBSCRIPTS
A subscript tells us how many of each atom there are

4
Multiple Choice
How many Sodium atoms are there in NaCl?
1
2
3
4
5
Multiple Choice
How many Magnesium atoms are there in Mg3N2?
1
2
3
4
6
Multiple Choice
How many Hydrogen atoms are there in Pentane, C5H12?
5
12
1
2
7
We CANNOT change the subscript, it would become something totally different!
Add a subscript of 2 after Oxygen and you make Hydrogen Peroxide.

8
Coefficiant
The large number in FRONT of the symbol. You have to multiply this number by all of the subscripts.
4 Iron Atoms
6 Oxygen Atoms

9
Multiple Choice
How many Hydrogens are in 2H2O?
2
4
6
1
10
Multiple Choice
How many Oxygens are in 6H2O?
2
12
6
11
Multiple Choice
How many Carbons are in 3CO2?
3
6
2
12

13
Balanced Equations
Follow the Law of Conservation
Have the same number and type of atoms on both sides
They just get rearranged!

14
Multiple Choice
Balanced or Not?
H2 + Cl2 ---> 2HCl
Balanced
Unbalanced
15
H2 + O2 ---> H2O
The hydrogen are balanced, but the oxygens are not.
You CAN change the coefficient.
You CANNOT change the subscript.
We put a two in front of the water and this balances the oxygen.
H2 + O2 ---> 2H2O

16
But....
H2 + O2 ---> 2H2O
Reactants: 2H, 2O Products: 4H, 2O
This causes the hydrogen to become unbalanced.
To fix this, we place a two in front of the hydrogen on the left side.
2H2 + O2 ---> 2H2O
17
2H2 + O2 ---> 2H2O
It's balanced now!
4 H and 2 O

18
Multiple Choice
Balanced or Not?
H2 + O2 ---> H2O
Balanced
Not
19
Multiple Choice
Balanced or not?
N2+6H2 ----> 2NH6
Balanced
Unbalanced
20
Multiple Choice
Which of the following equations is balanced?
P4 + 3O2 ---> 2P2O3
N4 + 3F2 ---> 2NF3
2LiF + Br2 ---> 2LiBr + F4
21
Multiple Choice
22
Multiple Choice

In this reaction, how much CO2 is produced?
42 g
24 g
23 g
No grams produced
23
Multiple Choice
Does this equation follow the Law of Conservation of Mass?
2Fe2O3 + 3C → 4Fe + 3 CO2
No, because there are more reactants than products.
No, because there are more products than reactants.
Yes, because the reactants equal the products.
Yes, because the reactants do not equal the products.
24
Multiple Choice
O + H2 → H2O
32 g of Oxygen completely reacts with 8 g of Hydrogen to produce _____ g of water.
8
12
24
40
25
Multiple Choice
Mg + F → MgF
24 g of magnesium reacts with 38 g of fluorine to produce _____ g magnesium fluoride.
26
27
8.5(E)
Investigate how evidence of chemical reactions indicates that new substances with different properties are formed and how that relates to the law of conservation of mass.
28
Multiple Choice

What is the symbol for Iron?
Ir
In
Fe
FE
29
Subscripts
The small number written directly behind the symbol of an element within a chemical formula.
It represents the number of atoms of that element.
If you DO NOT see a subscript it means there is ONLY ONE atom.

30
Coefficients
You must distribute the coefficient by multiplying it with each subscript.
REMEBER,if there are parenthesis you must multiply the subscript by all the subscripts (INSIDE) the parenthesis.
THEN multiply all your atoms by the coefficient.

31
Multiple Choice

How many Aluminum atoms are in Al2O3?
3
2
5
1
32
Multiple Choice
How many atoms are there TOTAL in H2SO4 ?
6
5
7
3
33

34
Multiple Choice
A subscript is the small number below the element symbol that tells the number of _______ of that element.
atoms
valence electrons
protons
elements
35
Multiple Choice
36
Physical Change
ONLY changes the form or the appearance of a substance.
Does not change or form a new substance.
Phase Changes

37

38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Chemical Changes
Substance is transformed into one or more different substances, such as when iron becomes rust.
IRREVERSIBLE - Cannot change it back to its original form.
A new product is produced.

43

44
Multiple Choice

45
Multiple Choice

46
Multiple Choice
47
Multiple Choice
48
Multiple Choice

49
Chemical Reactions
a process in which atoms of one or more substances (reactants) are rearranged to form one or more new substances (products).
Chemical changes are the result of chemical reactions.

50
Indicators of a Chemical Reaction:
COLOR CHANGE
GAS PRODUCTION/ Bubbles
LIGHT PRODUCTION
TEMPERTURE CHANGE
ODOR
NEW substances
PRECIPITATE

51
Open Ended
In your own words, tell me what is a precipitate and how is it formed?
52
Multiple Choice

Identify what type of change happening in the picture below...
Chemical Reaction
Physical Change
Change in State
Change in appearance only
53
Multiple Choice
The formation of a solid when two liquids combine is called?
Solidification
Freezing
Precipitate
Solubility
54
Multiple Choice
A Chemical Change is also known as what?
Physical Change
Physical Property
Chemical Reaction
Chemical Property
55
Multiple Choice
A green powder is heated & a gas is given off while it turns to a black solid. This describes
Physical Change
Chemical Change
Change in State
Change in Temperature
56
Multiple Choice
During Chemical Reactions, the new substances created are called?
Precipitate
Corrosion
Reactants
Products
57
Multiple Choice
During Chemical Reactions, the substances that are used are called?
Precipitate
Corrosion
Reactants
Products
58
Multiple Choice
59
Multiple Choice
A chemical reaction is balanced when
both sides have the same elements
Both sides have the same number of atoms
Same subscripts
Same coefficients
60
Multiple Choice
61
Multiple Choice
A chemical change is different than a physical change because in a chemical change
chemicals are used
molecules do not physically touch
a new substance is formed and in a physical no new substance is formed
the change can be seen but in a physical change it cannot
62
Types of Forces
63




Contact forces: These require physical
contact between objects.
Friction
Applied Forces
Normal Forces
64
Multiple Choice
Two object must touch each other for a contact force to have happened
yes
no
65


Friction
• Friction is a force that slows motion when two objects
rub against each other
• Friction opposes motion
66
Multiple Choice
Friction ______ Motion.
helps motion
opposes (slows) motion
67




●rubbing both the hands
together to create heat
●a child sliding down through a
slide in a park
●a book sliding across a table
●a book resting on a table
●a car parked on a hill
●a ladder leaning against a
wall
●A moving car
●rolling a soccer or tennis ball
●a marble rolling on the floor
68
Open Ended
Give one example of friction
69




Applied Force
A force that is applied to an object by
another object or person
• when you push a door open
• kick a ball
• pull a drawer out
70
Multiple Choice
An example of applied force is _______.
rain falling
earth orbiting
kicking a ball
71



Normal Forces
Upward push of a surface on an object
that keeps the object from falling through.
● Books sitting on a table
● A person sitting on a tabletop
72
Multiple Choice
Normal forces
make objects fly
keep objects from falling through surfaces
give objects speed
73
Open Ended
Give and example of a normal force.
74





Non-contact forces: These act on objects
even if they are not physically touching.
Gravity
Magnetism
75




Gravitational Force
The attraction between two objects
with mass, such as Earth pulling objects
toward its center.
● Rain falling to Earth
● Tides in the ocean
● Earth’s revolution around the sun
76
Drag and Drop
77
Net Force review
78

As a review- Net force is the combination of forces on an object.
Answer will need the netforce (in Newtons) and the direction
Net Force
79
Multiple Choice
What is Force?
A pull exerted on an object
A push exerted on an object
A push or pull exerted on an object
Nothing
80
Multiple Choice
N (Newtons)
lbs (pounds)
g (grams)
m/s (meters per second)
81
As a Review:
If the forces are going in the same direction: you add the forces
If the directions are going in different directions: you subtract the forces
If there is a combination: add the ones that are the same, then subtract the different directions.
82
Multiple Choice

How do you find the net force of this image.
Add up all the arrows
Add a green arrow and the orange arrow.
Subtract all the arrows.
Add the green arrows and subtract the orange arrow.
83
Multiple Choice
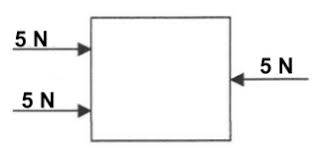
Is the Net Force balanced or Unbalanced?
Balanced
Unbalanced
84
Multiple Choice

What is the Overall Net Force and which side has a greater Net Force?
12, Right
0, Both are balanced
12, Both are balanced
0, Left
85
Multiple Choice

What is the net force and to what direction.
210 N to the right
280 N to the right
110 N to the left
180 N to the left
86
Multiple Choice
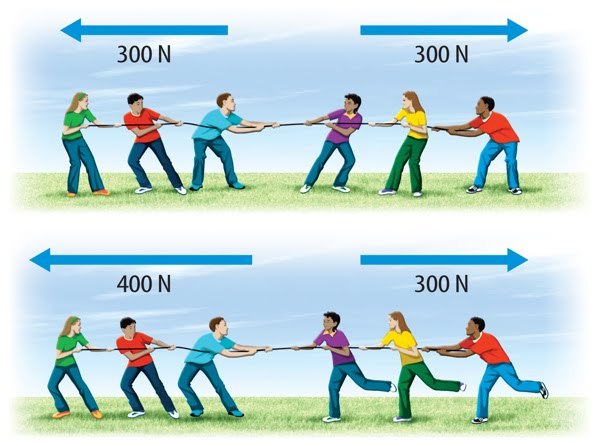
Which one is balanced?
top
bottom
both
Neither is balanced
87
Multiple Choice
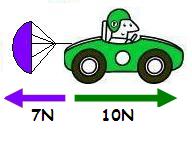
What is the net force?
17 N Right
3 N Right
3 N Left
17 N Left
88
Multiple Choice

Which direction will the object move?
Up
Down
Right
Left
89
Multiple Choice

What is the net force?
65 N left
65 N right
35 N left
35 N right
90
Multiple Choice
If the wind is heading south with a force of 5 N, and a car is going north with force of 78936 N, what is the Net Force and Direction.
78931 N North
78931 N South
78531 N North
78531 N South
91
Multiple Choice
If a car has a force of 54 N forwards and the brakes have a force of 73 N backwards, what is the overall Net Force.
16 backwards
19 forwards
18 backwords
23 forwards
92
Multiple Choice
If a boat traveling East has a Net Force of 324 N, and the wind blowing West has a Net Force of 789 N. What is the overall Net Force and which side?
465 N, East
465 N, West
1113 N, West
1113 N, East
93
Newton's Laws of Motion

94

95

96
Multiple Choice
97
Multiple Choice
98
Multiple Choice
99
Multiple Choice
100
Multiple Choice

101
Multiple Choice

102

103

104
Multiple Choice
What is Newton's 2nd Law?
Every action has an equal and opposite reaction
An object at rest stays at rest, an object in motion stays in motion.
F = ma
Friction
105
Multiple Choice
106
Multiple Choice
107
Multiple Choice
108
Multiple Choice
You push on a tree with 20 N of force. If the tree doesn't move, the tree is pushing back on you with _____of force.
20N
10N
40N
70N
109
Multiple Choice
The mass of a wagon is 40 kg. The wagon accelerates at 8 m/s2 . What is the force?
38.25 Kg
320N
5N
3.825Kg
110
Also known as the "Law of Action/Reaction"

111

112
Multiple Choice
113
Multiple Choice
114
Multiple Choice
Two students with different masses are sitting in roll-y chairs. The lighter one uses his feet to push the other
The lighter student will move faster because he has less mass
The heavier student will move more because he is being pushed
They will move with the same speed
The heavier student shouldn't move at all
115
Multiple Choice
When you hit a tennis ball with a racket...
The ball exerts more force on the racket
The ball and racket exert the same force on each other
The racket exerts more force on the ball
It depends on how hard you hit it
Checkpoint 2 Review
8th Grade Science

Show answer
Auto Play
Slide 1 / 115
SLIDE
Similar Resources on Wayground

106 questions
Animal Ecology
Presentation
•
6th - 8th Grade

106 questions
Ecology Video Lesson
Presentation
•
6th - 8th Grade

110 questions
Rivervalley Civilizations
Presentation
•
6th - 7th Grade

114 questions
PHYSICAL vs POLITICAL MAPS
Presentation
•
6th - 8th Grade

109 questions
Similarity Test
Presentation
•
8th Grade

109 questions
Introduction to Similarities in Math
Presentation
•
8th Grade

111 questions
FINAL Study Guide (Scientific Method,)
Presentation
•
6th - 8th Grade

107 questions
Walk through Geologic Time
Presentation
•
7th - 9th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

18 questions
Oceans, Atmosphere, Climate Vocabulary Amplify
Quiz
•
6th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade