

Colligative Properties
Presentation
•
Chemistry
•
University
•
Easy
+2
Standards-aligned
Genia Harris
Used 3+ times
FREE Resource
14 Slides • 26 Questions
1

2

3
Multiple Choice
Which of the following is NOT a colligative property of a solution?
Freezing point
Boiling point
Osmotic pressure
Density
4
Open Ended
Why do you think understanding colligative properties is important in real-life situations?
5

6
Multiple Choice
Which of the following is NOT a colligative property of solutions?
Vapor-pressure lowering
Boiling-point elevation
Freezing-point lowering
Density increase
7

8
Open Ended
Explain how the presence of a solute affects the evaporation of a solvent in a solution.
9

10
Multiple Select
Which of the following statements about freezing point depression and boiling point elevation are correct?
The freezing point of a solution is higher than that of the pure solvent.
The boiling point of a solution is higher than that of the pure solvent.
The freezing point of a solution is lower than that of the pure solvent.
The boiling point of a solution is lower than that of the pure solvent.
11

12
Fill in the Blanks
Type answer...
13

14
Open Ended
Describe the process of osmosis and the role of osmotic pressure in this process.
15

16
Multiple Choice
What is the function of the semipermeable membrane in the process of osmosis as shown in the diagram?
It allows solute particles to pass through but not solvent molecules.
It allows solvent molecules to pass through but not solute particles.
It prevents both solvent and solute particles from passing through.
It allows both solvent and solute particles to pass through freely.
17

18
Multiple Choice
Which of the following is a reason why salt is spread on icy roads?
To increase the boiling point of water
To melt the ice by lowering the freezing point
To make the roads rougher
To prevent rust on vehicles
19

20
Open Ended
How do colligative properties help us understand the behavior of solutions in everyday life?
21
Multiple Choice
Which of the following is a characteristic of colligative properties?
They depend on the number of dissolved solute particles.
They depend on the type of solute particle.
They are the same as the properties of the solvent.
They do not affect freezing or boiling points.
22
Open Ended
Explain how the addition of solutes to water is applied in real-life situations, such as those mentioned in the slides.
23

24

25

26

27
Multiple Choice

The table below gives information about four aqueous solutions at standard pressure.
Which list of solutions is arranged in order from highest boiling point to lowest boiling point?
A, B, D, C
A, C, B, D
C, D, B, A
D, B, C, A
28
Multiple Choice

What is the vapor pressure of propanone at 50C?
37 kPa
50 kPa
83 kPa
101 kPa
29
Multiple Choice

Definition of normal boiling point:
The boiling point of a substance at an elevation of 8,000
The boiling point of a substance of 0.61 atm
The boiling point of a substance at standard pressure
The boiling point at the most highest elevation available
30
Multiple Choice
What is the boiling point of a solution that contains 7.6 moles of CH4 in 0.850kg of water? Kbp= 0.512 degrees C/m
4.58 degrees C
100 degrees C
104.58 degrees C
31
Multiple Choice
Which law specifically governs the relative lowering of vapor pressures in solutions?
a) van’t Hoff law
b) Boyle’s law
c) Raoult’s law
d) Amagat’s law
32
Multiple Choice
33
Multiple Choice
Find the boiling point of a solution containing 15.0 g sucrose (molar mass = 342.3g/mol), in 100g of water. (Kb = 0.512 oC/m)
100.2
99.8
0.2
-0.2
34
Multiple Choice

35
Multiple Choice
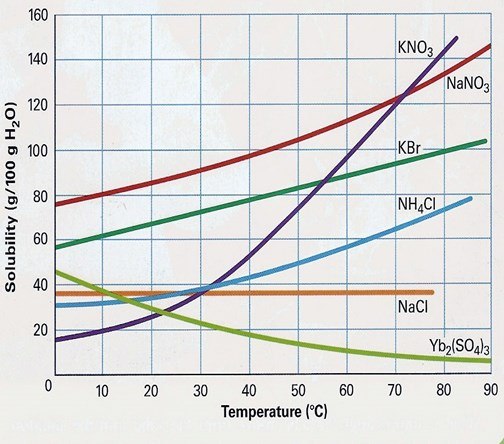
At which temperature do KBr and KNO3 have the same solubility?
60
55
50
Never
36
Multiple Choice

37
Multiple Choice

38
Multiple Choice

39
Multiple Choice

40
Multiple Choice


Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

35 questions
SV Agreement
Presentation
•
University

36 questions
EV3U8
Presentation
•
University

32 questions
Hindi Shabdavali
Presentation
•
KG

37 questions
Introduction to Ecclesiology PPT
Presentation
•
KG - University

35 questions
Chapter 6- Chemical Composition
Presentation
•
University

36 questions
Simple, Compound, and Complex Sentences
Presentation
•
University

36 questions
English 3 - UNIT 4 Cars and Driving
Presentation
•
University

35 questions
Phrasal Verbs
Presentation
•
University
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

20 questions
Disney Trivia
Quiz
•
University

24 questions
5th Grade Math EOG Review
Quiz
•
KG - University

14 questions
Reading- SC Ready Practice
Quiz
•
5th Grade - University

25 questions
APUSH Decades Review
Quiz
•
9th Grade - University

40 questions
Famous Logos
Quiz
•
7th Grade - University
![Repaso - La Calaca Alegre (whole book) [Twist]](https://cf.quizizz.com/image/image-loader.svg)
44 questions
Repaso - La Calaca Alegre (whole book) [Twist]
Quiz
•
9th Grade - University

14 questions
(5-3) 710 Mean, Median, Mode & Range Quick Check
Quiz
•
6th Grade - University