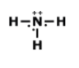
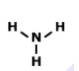
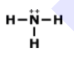
Lewis Structures
Quiz
•
Chemistry
•
10th Grade
•
Easy
ANA WU
Used 1+ times
FREE Resource
Enhance your content in a minute
20 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to the octet rule most elements need _______ valence electrons.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Hydrogen needs _____ electrons in its valence shell to be stable.
3.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
A covalent bond is
4.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

Which of the following is the correct Lewis dot structure for the molecule fluorine (F2)?
5.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
How many electrons can be used in the Lewis Structure for : NO3 -
6.
MULTIPLE CHOICE QUESTION
3 mins • 1 pt
How many valence electrons are available for bonding in the sulfate ion (SO4-2)?
7.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
How many valence electrons are in Phosphorus?
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

20 questions
Electron Arrangement of Atoms
Quiz
•
9th - 12th Grade

15 questions
Advance program 2020 yr 11 Chem Periodic Table Trends
Quiz
•
10th - 11th Grade

20 questions
Chemical Nomenclature
Quiz
•
10th - 11th Grade

16 questions
Electricity Quiz
Quiz
•
KG - University

19 questions
Equilibrium & Le Chatelier's Principle Review
Quiz
•
9th - 12th Grade

20 questions
atomic structure quiz
Quiz
•
10th Grade

21 questions
ELECTRON ARRANGEMENT
Quiz
•
10th - 12th Grade

20 questions
Chemistry Semestral Exam Review
Quiz
•
10th Grade
Popular Resources on Wayground

8 questions
2 Step Word Problems
Quiz
•
KG - University

20 questions
Comparing Fractions
Quiz
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

10 questions
Latin Bases claus(clois,clos, clud, clus) and ped
Quiz
•
6th - 8th Grade

22 questions
fractions
Quiz
•
3rd Grade

7 questions
The Story of Books
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

20 questions
Elements, Compounds, and Mixtures
Quiz
•
10th Grade

20 questions
Practice: E-Con, Orbital Notation, Noble Gas Notation
Quiz
•
10th Grade

20 questions
Stoichiometry Practice
Quiz
•
10th Grade

22 questions
Periodic Trends Freshman
Quiz
•
10th Grade

23 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

20 questions
Periodic Trends
Quiz
•
10th Grade

13 questions
Mole Conversions
Quiz
•
9th - 12th Grade