
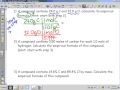
Moles and Stoichiometry: Empirical Formula Challenges
Interactive Video
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Medium
Mia Campbell
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What unit is used to convert grams of an element to moles in the dimensional analysis?
Liters per mole
Mole ratio
Percentage by mass
Grams per mole
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the gram formula mass (gfm) used for in the conversion process?
To calculate the yield of a reaction
To convert grams to moles
To determine the compound's density
To convert moles to liters
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does dimensional analysis help you find in the context of empirical formulas?
The concentration of solutions
The energy change per mole
The moles of each element
The volume of reactants
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What do you need to calculate after finding the moles of each element to determine the empirical formula?
Molecular weight
Percentage composition
Lowest whole number ratio
Total mass of the compound
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If given moles directly in a problem, what is the next step to find the empirical formula?
Assume a 100 gram sample
Divide by the lowest mole number
Find the molecular formula
Convert to grams
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the empirical formula written after determining the mole ratios?
As a balanced equation
Using molecular weights
With subscripts indicating mole ratios
As a percentage composition
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to divide by the lowest mole number when calculating empirical formulas?
To ensure the formula is as simple as possible
To balance the chemical equation
To calculate the reactant's purity
To determine the reaction rate
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
