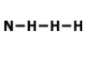Lewis Dot Structures - Covalent Bonds
Authored by Valerie Faculty
Chemistry
9th - 12th Grade
NGSS covered
Used 4+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
14 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
The type of bond where valence electrons are SHARED is called:
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
According to the octet rule most elements need _______ valence electrons.
Tags
NGSS.HS-PS1-2
NGSS.HS-PS1-1
3.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Hydrogen needs _____ electrons in its valence shell to be stable.
4.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

How many electrons does each bond line indicate are shared?
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Three pairs of electrons are shared in a
6.
MULTIPLE SELECT QUESTION
45 sec • 1 pt
What two types of atoms make a covalent bond? (Choose 2 answers)
A nonmetal and a metalloid
7.
MULTIPLE CHOICE QUESTION
10 mins • 10 pts

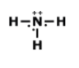
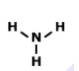
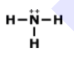
How many single bonds does this have?
1
2
3
4
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

10 questions
Quiz : Reaction of amide
Quiz
•
12th Grade

13 questions
OCR Chemistry A – 5.2.3(a-d) Redox Titrations
Quiz
•
12th Grade

16 questions
Chemistry Diagnostic Pre-Test
Quiz
•
9th Grade

10 questions
XI Chemistry Unit - 2 Part - 4A
Quiz
•
11th Grade

10 questions
Goc 1
Quiz
•
11th - 12th Grade

10 questions
Ionic Bond
Quiz
•
10th Grade

15 questions
Tspé.04. Transformation acide-base et pH
Quiz
•
12th Grade

16 questions
Chemical Reactions
Quiz
•
9th - 10th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

30 questions
Energy Review
Quiz
•
9th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

20 questions
Chemistry: Classification of Matter
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade