
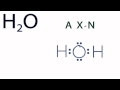
Molecular Geometry of Water
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in determining the molecular geometry of H2O?
Using a 3D model
Consulting a table
Examining the Lewis structure
Applying the AXN notation
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to the valence shell electron pair repulsion theory, how do electron pairs arrange themselves?
As far apart as possible
In a circular pattern
In a linear fashion
As close as possible
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What shape does the water molecule have when visualized in three dimensions?
Linear
Bent
Trigonal planar
Tetrahedral
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the AXN notation, what does 'A' represent?
The central atom
The bond angle
The number of attached atoms
The number of lone pairs
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many non-bonding electron pairs are present in the water molecule according to the AXN notation?
Four
Three
Two
One
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molecular geometry of H2O as determined by the AXN notation?
Trigonal planar
Tetrahedral
Linear
Bent
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What bond angle is associated with the bent molecular geometry of water?
90°
120°
109.5°
180°
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
