
Molecular Geometry and Polarity of H2S
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Olivia Brooks
FREE Resource
Read more
7 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What initial observation is made about the Lewis structure of H2S?
It has no lone pairs.
It is a linear molecule.
It appears symmetrical.
It appears asymmetrical.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What theory is used to understand the arrangement of electrons in H2S?
Molecular Orbital Theory
Valence Bond Theory
Crystal Field Theory
Valence Shell Electron Pair Repulsion Theory
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
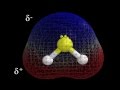
In the molecular geometry of H2S, what color represents sulfur?
Red
Blue
Yellow
White
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the lone pairs of electrons affect the shape of the H2S molecule?
They have no effect.
They make it linear.
They push the hydrogen atoms upward.
They push the hydrogen atoms downward.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What indicates that H2S is a polar molecule?
It has no poles.
It has two positive poles.
It has two negative poles.
It has a positive and a negative pole.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the blue and red colors in the polarity explanation of H2S?
Both are neutral.
Blue is negative, red is positive.
Blue is positive, red is negative.
Both are positive.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is suggested as a better way to draw the Lewis structure of H2S?
As a linear molecule
With lone pairs at the bottom
With no lone pairs
Based on its molecular geometry
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
