
Classifying Properties of Matter
Interactive Video
•
Science
•
6th - 10th Grade
•
Practice Problem
•
Hard
Standards-aligned
Ethan Morris
FREE Resource
Standards-aligned
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
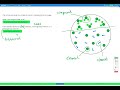
What is the main task in the given problem?
To measure the size of the sample
To find the temperature of the sample
To determine the composition and classification of the sample
To identify the color of the sample
Tags
NGSS.MS-PS1-1
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many different components are identified in the sample?
Four
Two
Three
One
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first component identified as?
A compound
A molecule
A mixture
An element
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What distinguishes the second component from the first?
It is a compound made of multiple elements
It is larger in size
It is a different color
It is a single atom
Tags
NGSS.MS-PS1-1
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the second component identified as?
A molecule
A mixture
A compound
An element
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is unique about the third component?
It is a different color
It is a mixture
It consists of two identical atoms
It is a compound
Tags
NGSS.MS-PS1-1
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many elements and compounds are present in the sample?
Two elements and one compound
One element and two compounds
Two compounds and one element
Three elements
Tags
NGSS.MS-PS1-1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
