
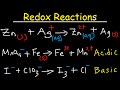
Balancing Redox Reactions: Acidic and Basic Solutions
Interactive Video
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Medium
Sophia Harris
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a redox reaction, if the left side has a higher charge, where are electrons added?
Equally on both sides
Not added at all
To the side with the higher charge
To the side with the lower charge
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What indicates that a half-reaction is an oxidation process?
No electrons are involved
Electrons are on the right side
Electrons are on the left side
Only H2O is added
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is added to the left side of a redox reaction under acidic conditions to balance oxygen atoms?
H+ ions
H2O molecules
OH- ions
Electrons
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you balance hydrogen atoms in acidic conditions?
Add H2O molecules
Add electrons
Add H+ ions
Add OH- ions
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of multiplying equations in redox balancing?
To increase the reaction rate
To make the number of electrons equal
To decrease the solution's pH
To balance the number of molecules
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the least common multiple used for in redox reaction balancing?
To determine the amount of solvent
To calculate the final charge
To balance the atomic masses
To equalize electron counts in half-reactions
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Under basic conditions, what is added to balance the charges in a redox reaction?
Electrons
H2O molecules
OH- ions
H+ ions
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
