
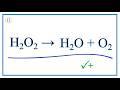
Balancing Chemical Equations Concepts
Interactive Video
•
Chemistry, Science
•
8th - 10th Grade
•
Practice Problem
•
Medium
Sophia Harris
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial count of hydrogen atoms on the reactant side of the equation H2O2 yields O2 plus H2O?
4
3
2
1
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many oxygen atoms are initially present on the product side before balancing?
3
4
5
2
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the strategy used to balance an odd number of oxygen atoms?
Add more hydrogen
Multiply by 2
Divide by 2
Subtract oxygen
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
After placing a 2 in front of H2O, how many hydrogen atoms are on the product side?
3
4
2
5
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the new count of oxygen atoms on the product side after adjusting H2O?
5
4
3
2
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What coefficient is placed in front of H2O2 to balance the equation?
1
2
3
4
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many hydrogen atoms are on each side after balancing the equation?
4
5
3
2
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
