
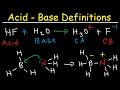
Bronsted-Lowry and Lewis Concepts
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Medium
Jackson Turner
Used 12+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to the Arrhenius definition, what do acids release in a solution?
Hydroxide ions
Hydronium ions
Sodium ions
Chloride ions
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of an Arrhenius base?
HCl
H2SO4
NaOH
HNO3
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the Bronsted-Lowry definition, what is a proton donor called?
Lewis acid
Bronsted-Lowry acid
Arrhenius base
Bronsted-Lowry base
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the conjugate base of HCl in a Bronsted-Lowry reaction?
H2O
Cl-
H3O+
OH-
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you determine the conjugate acid of a molecule?
Add a hydroxide ion
Remove a proton
Add a proton
Remove an electron pair
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the conjugate base of NH3?
NH4+
NH2-
NH3+
NH4-
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the reaction between carbonate and water, what is the Bronsted-Lowry base?
Bicarbonate
Carbonate
Water
Hydroxide
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
