
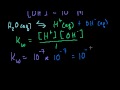
Understanding Water Chemistry and Autoionization
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond is responsible for holding water molecules together in liquid and solid states?
Hydrogen bond
Metallic bond
Covalent bond
Ionic bond
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the process called when water molecules spontaneously ionize to form hydronium and hydroxide ions?
Hydrolysis
Autoionization
Neutralization
Condensation
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the autoionization of water, what is the equilibrium constant (Kw) at room temperature?
10^-7
10^-10
10^-3
10^-14
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the 'p' in pH and pKw stand for in chemistry?
Potential
Power
Partial
Pressure
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the pH of pure water at room temperature?
3
7
14
10
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does an increase in hydrogen ion concentration affect the pH of a solution?
Decreases pH
Neutralizes pH
Increases pH
No effect on pH
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the pOH of pure water at room temperature?
14
10
7
3
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
