
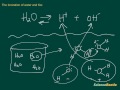
Ionization of Water Concepts
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of the video tutorial?
The chemical reactions of hydrogen
The ionization of water and the water constant KW
The structure of the water molecule
The properties of acids and bases
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the ionization of water produce?
Hydrogen ions and hydroxide ions
Carbon dioxide and water
Oxygen and hydrogen gas
Salt and water
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the water molecule considered polar?
Because it is a gas at room temperature
Because it has a positive and a negative end
Because it forms a solid at low temperatures
Because it is a non-conductor of electricity
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when another water molecule interacts with a water molecule in solution?
It releases oxygen gas
It becomes a non-polar molecule
It forms a solid precipitate
It forms hydrogen and hydroxide ions
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of the equilibrium process in the ionization of water?
The solution becomes basic
The solution becomes acidic
The water completely ionizes
The rate of ion formation equals the rate of water formation
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the ionization constant KW represent?
The ratio of the concentrations of hydrogen and hydroxide ions
The difference between the concentrations of hydrogen and hydroxide ions
The sum of the concentrations of hydrogen and hydroxide ions
The product of the concentrations of hydrogen and hydroxide ions
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the value of the ionization constant KW at 25°C?
1 * 10^-14
1 * 10^-7
1 * 10^-12
1 * 10^-10
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
