
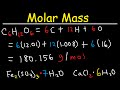
Molar Mass and Formula Weight Concepts
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Easy
Ethan Morris
Used 9+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the atomic mass of carbon as mentioned in the video?
44.01
12.01
6
16
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the molar mass of CO2?
Subtract the atomic mass of oxygen from carbon
Multiply the atomic mass of carbon by two
Add the atomic masses of one carbon and two oxygen atoms
Divide the atomic mass of carbon by the atomic mass of oxygen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of methane (CH4) as calculated in the video?
44.01 g/mol
58.44 g/mol
18.016 g/mol
16.042 g/mol
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula weight of sodium chloride (NaCl)?
39.998 g/mol
58.44 g/mol
18.016 g/mol
342.296 g/mol
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is true about formula weight?
It is only used for molecular compounds
It is not related to the periodic table
It is calculated by subtracting atomic masses
It is the same as molar mass for ionic compounds
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of glucose (C6H12O6) as calculated in the video?
342.296 g/mol
525.992 g/mol
219.076 g/mol
180.156 g/mol
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many carbon atoms are in sucrose (C12H22O11)?
6
11
12
22
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
