
Acidity and Conjugate Bases in Organic Chemistry
Interactive Video
•
Chemistry
•
10th Grade - University
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which molecule is more acidic based on pKa values?
Both have the same acidity
Acetic acid
Ethanol
Neither is acidic
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the conjugate base of ethanol called?
Methoxide
Hydroxide
Ethoxide
Acetate
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the acetate ion more stable than the ethoxide ion?
It has more hydrogen atoms
It is less electronegative
It has a higher pKa value
It has a carbonyl group that withdraws electron density
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What effect does the carbonyl group have on the acetate ion?
It has no effect
It increases the positive charge on the oxygen
It withdraws electron density from the oxygen
It donates electrons to the oxygen
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
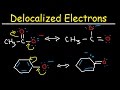
Which molecule is more acidic, cyclohexanol or phenol?
Neither is acidic
Phenol
Both are equally acidic
Cyclohexanol
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the conjugate base of phenol called?
Ethoxide ion
Phenoxide ion
Cyclohexoxide ion
Acetate ion
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the phenoxide ion more stable than the cyclohexoxide ion?
It is less electronegative
It has more resonance structures
It has fewer resonance structures
It has a higher pKa value
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
