
Understanding Nitration of Aromatic Compounds
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Ethan Morris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of the methyl group in the nitration of toluene?
It prevents any reaction from occurring.
It deactivates the benzene ring.
It acts as an ortho-para director.
It directs the nitro group to the meta position.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
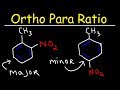
In the nitration of toluene, what is the approximate ratio of ortho to para products?
60% ortho, 40% para
70% ortho, 30% para
50% ortho, 50% para
40% ortho, 60% para
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the bulkiness of the ethyl group affect the ortho-para ratio in ethyl benzene nitration?
It prevents the reaction from occurring.
It favors the para product.
It favors the ortho product.
It results in a 50/50 ortho-para ratio.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the ortho-para ratio when a bulky tert-butyl group is used?
The reaction does not occur.
The ratio remains 50/50.
The para product is favored.
The ortho product is favored.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the presence of a tert-butyl group, what is the expected yield of the para product?
80%
20%
40%
60%
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a benzene ring with both tert-butyl and methyl groups, which position is more accessible for the nitro group?
Ortho to methyl
Ortho to tert-butyl
Para to tert-butyl
Meta to both groups
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When tert-butyl and methyl groups are ortho to each other, which site is most accessible for nitration?
Site B
Site A
All sites are equally accessible
Site C
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
