
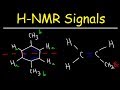
Understanding HMR and CNMR Spectra
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What determines the number of signals in an HMR spectrum for a molecule?
The number of chemically equivalent hydrogen atoms
The molecular weight of the compound
The number of carbon atoms
The presence of double bonds
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the HMR spectrum of propane, how many signals are expected?
Two
One
Four
Three
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why do the methyl groups in dimethyl ether show up as one signal in the HMR spectrum?
They are part of a double bond
They are in different chemical environments
They are in the same chemical environment
They are attached to different carbon atoms
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many signals are expected in the HMR spectrum of 2-methylpentane?
Six
Four
Three
Five
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of signals in the HMR spectrum of benzene?
Three
Four
Two
One
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In bromobenzene, how many signals are observed in the CNMR spectrum?
Two
Four
Three
Five
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many HMR signals are expected for para xylene?
One
Two
Three
Four
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
