
Kinetic Energy and Temperature: Exploring Their Interconnectedness
Interactive Video
•
Physics, Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula for kinetic energy?
m times v
mv squared
one half mv squared
m plus v squared
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is temperature related to kinetic energy?
Temperature is inversely proportional to kinetic energy
Temperature is proportional to the average kinetic energy of molecules
Temperature is unrelated to kinetic energy
Temperature is proportional to the square of kinetic energy
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
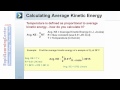
What is the formula for average kinetic energy?
one half mv squared
R plus T
three halves RT
RT squared
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which temperature scale is based on absolute zero?
Celsius
Rankine
Fahrenheit
Kelvin
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the temperature at which molecules have no kinetic energy?
0 Celsius
0 Fahrenheit
100 Kelvin
0 Kelvin
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you convert Celsius to Kelvin?
Divide by 273
Multiply by 273
Subtract 273
Add 273
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the freezing point of water in Fahrenheit?
212 degrees
0 degrees
32 degrees
100 degrees
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Popular Resources on Wayground

8 questions
Spartan Way - Classroom Responsible
Quiz
•
9th - 12th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

3 questions
Integrity and Your Health
Lesson
•
6th - 8th Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

9 questions
FOREST Perception
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade
Discover more resources for Physics

10 questions
Exit Check 4.5 - Impulse
Quiz
•
9th Grade

20 questions
Simple Machines and Mechanical Advantage Quiz
Quiz
•
9th Grade

20 questions
Electricity/Magnets
Quiz
•
9th Grade

20 questions
Light Vs. Sound Waves
Quiz
•
9th - 12th Grade

14 questions
Bill Nye Waves
Interactive video
•
9th - 12th Grade

37 questions
CIA #3 Study Guide: ACPS
Quiz
•
10th Grade

20 questions
Waves
Quiz
•
9th - 12th Grade

10 questions
Exploring the Properties of Waves
Interactive video
•
9th - 12th Grade