
Calorimetry Concepts and Applications
Interactive Video
•
Physics, Chemistry, Science
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of the video?
The history of physics
Chemical reactions
The structure of atoms
Calorimetry and its applications
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
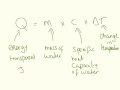
What is a calorie defined as in the context of calorimetry?
The energy required to heat one liter of water by ten degrees
The energy required to heat one liter of water by one degree
The energy required to heat one gram of water by one degree
The energy required to heat one gram of water by ten degrees
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which unit is used to measure the energy transferred in the discussed equation?
Kilowatts
Joules
Watts
Calories
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the specific heat capacity of water as mentioned in the video?
4.2 calories per liter degree
4.2 joules per gram degree
4.2 calories per gram degree
4.2 joules per liter degree
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the symbol 'delta' represent in the equation?
Change in mass
Change in volume
Change in energy
Change in temperature
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a common source of error in the calorimetry experiment?
Using too much water
Measuring temperature in Fahrenheit
Not using a lid
Using a metal container
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why might the results of the experiment not be completely accurate?
The experiment is done too quickly
The water is not pure
Energy is lost to the surroundings
The water is too cold
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
