
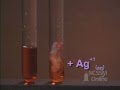
Chemical Equilibrium and Reaction Effects
Interactive Video
•
Chemistry, Science, Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does L's principle suggest about a reaction at equilibrium when stress is applied?
The reaction will slow down.
The reaction will speed up.
The reaction will shift to relieve the stress.
The reaction will stop.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the color of the solution when iron(III) nitrate is added?
It turns blue.
It becomes colorless.
It turns a darker red.
It turns green.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the addition of potassium thiocyanate affect the reaction?
The reaction becomes colorless.
The reaction stops.
The reaction shifts in the forward direction.
The reaction shifts in the reverse direction.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the effect of adding sodium hydroxide to the reaction mixture?
The red color intensifies.
The red color disappears completely.
The solution turns blue.
The red color decreases significantly.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when potassium chloride is added to the solution?
The color intensity increases.
The color intensity decreases.
The solution turns green.
The solution becomes colorless.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is observed when silver nitrate is added to the reaction mixture?
The solution turns blue.
The red color disappears and a white solid forms.
The red color intensifies.
The solution becomes colorless.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the formation of a white solid indicate when silver nitrate is added?
Formation of a new complex ion.
Precipitation of iron ions.
Formation of slightly soluble silver thiocyanate.
Complete dissolution of reactants.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
