
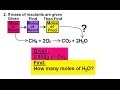
Moles and Reactions of Methane
Interactive Video
•
Chemistry, Science, Mathematics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction occurs when methane is burned in the presence of oxygen?
Synthesis reaction
Single replacement reaction
Decomposition reaction
Combustion reaction
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in determining the moles of water produced from methane?
Convert the mass of methane to moles
Measure the temperature change
Calculate the volume of methane
Determine the pressure of the system
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many hydrogen atoms are present in one molecule of methane?
Three
Four
One
Two
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of methane (CH4)?
20.00 g/mol
18.02 g/mol
16.04 g/mol
12.01 g/mol
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the ratio of moles of water produced to moles of methane used in the reaction?
2:1
3:1
1:2
1:1
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If you start with 0.0425 moles of methane, how many moles of water will be produced?
0.02125 moles
0.0425 moles
0.0850 moles
0.1700 moles
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final step in calculating the moles of water produced?
Convert moles of water to grams
Subtract moles of methane from water
Multiply moles of methane by the reaction ratio
Add moles of methane and water
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
