
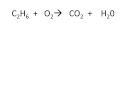
Balancing Hydrocarbon Combustion Reactions
Interactive Video
•
Chemistry, Science, Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary goal when balancing hydrocarbon combustion reactions?
To reduce the number of reactants
To increase the number of products
To make sure the number of atoms for each element is equal on both sides
To ensure the equation is aesthetically pleasing
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the combustion of methane, what is the first element you should balance?
Hydrogen
Nitrogen
Carbon
Oxygen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many hydrogens are on the left side of the methane combustion equation?
Six
Four
Two
Eight
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When balancing propane combustion, how many carbon atoms are on the left side?
One
Three
Four
Two
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the coefficient for water when balancing hydrogen in propane combustion?
Five
Two
Three
Four
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many total oxygen atoms are needed on the right side of the propane combustion equation?
Eight
Ten
Fourteen
Twelve
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a common issue when balancing reactions with an odd number of hydrogens?
It results in an odd number of oxygens
It makes the reaction impossible to balance
It requires more reactants
It leads to an excess of products
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
