
Chemical Bonding Concepts and Electronegativity
Interactive Video
•
Chemistry, Science, Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why do atoms form chemical bonds?
To increase their potential energy
To become more stable by reducing potential energy
To increase their atomic number
To exist alone in nature
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are valence electrons?
Electrons in the innermost energy level
Electrons involved in chemical bonding
Protons in the nucleus
Neutrons in the nucleus
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In an ionic bond, what happens to the electrons?
They remain in the same atom
They are lost to the environment
They are transferred from one atom to another
They are shared equally between atoms
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of an ionic bond?
CH4
O2
NaCl
H2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
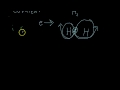
What is the main characteristic of covalent bonding?
Proton sharing
Electron sharing
Neutron transfer
Electron transfer
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond is formed when electrons are shared equally?
Ionic bond
Polar covalent bond
Non-polar covalent bond
Metallic bond
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is electronegativity used to determine bond type?
By multiplying the electronegativities of two elements
By subtracting the electronegativities of two elements
By dividing the electronegativities of two elements
By adding the electronegativities of two elements
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
