
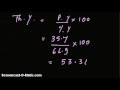
Mole Ratios and Yield Calculations
Interactive Video
•
Chemistry, Mathematics, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relationship between theoretical yield, practical yield, and percent yield?
Theoretical yield is the product of practical yield and percent yield.
Theoretical yield is the sum of practical yield and percent yield.
Theoretical yield is the quotient of practical yield and percent yield.
Theoretical yield is the difference between practical yield and percent yield.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If the practical yield is 35.7 and the percent yield is 66.9, what is the theoretical yield?
2.38
3.34
4.56
5.67
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you convert grams to moles?
Multiply the mass by the molar mass.
Divide the mass by the molar mass.
Add the mass to the molar mass.
Subtract the molar mass from the mass.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the mole ratio of AlBr3 to Br2 in the given reaction?
1:1
2:3
1:2
3:2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of establishing a mole ratio in a chemical reaction?
To find the amount of product formed.
To balance the chemical equation.
To calculate the theoretical yield.
To determine the limiting reactant.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If 2 moles of AlBr3 produce 3 moles of Br2, how many moles of Br2 are produced from 0.2 moles of AlBr3?
0.6
0.5
0.4
0.3
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of bromine used in the calculation?
27 g/mol
36 g/mol
18 g/mol
9 g/mol
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
