
Naming and Formulas of Ionic Compounds
Interactive Video
•
Chemistry, Science
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary purpose of a chemical formula?
To provide a recipe for making a compound
To explain the history of a compound
To describe the taste of a compound
To list the colors of a compound
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In binary ionic compounds, what happens to the name of the non-metal?
It is written in uppercase
It is replaced with a number
It changes to an 'ide' ending
It keeps its original name
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the key characteristic of ionic compounds that the crisscross method helps to achieve?
The total charges must be zero
The total charges must be positive
The total charges must be equal to the atomic number
The total charges must be negative
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in the crisscross method?
Calculate the molecular weight
Write the elements with their charges in superscript
Draw a diagram of the compound
List the elements alphabetically
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When using the crisscross method, what should you do if the subscript is one?
Write it as zero
Ignore it
Double it
Underline it
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
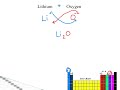
What is the correct chemical formula for a compound formed between lithium and oxygen?
LiO2
Li2O2
Li2O
LiO
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the name of the compound with the formula CaCl2?
Calcium chlorate
Calcium chlorite
Calcium dichloride
Calcium chloride
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
