
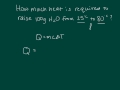
Heat Transfer and Temperature Change
Interactive Video
•
Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary equation used to calculate the heat required to change the temperature of a substance?
P = IV
Q = mcΔT
E = mc^2
F = ma
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When considering a temperature change from 25°C to 80°C, what phase is water in?
Solid
Liquid
Gas
Plasma
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the specific heat capacity of water used in the example problem?
3.18 J/g°C
4.18 J/g°C
2.18 J/g°C
1.00 J/g°C
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the change in temperature (ΔT) in the equation Q = mcΔT?
Average of initial and final temperatures
Sum of initial and final temperatures
Final temperature minus initial temperature
Initial temperature minus final temperature
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the heat required to raise 100 grams of water from 25°C to 80°C?
229.9 J
2,299 J
22.99 J
22,990 J
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the context of the video, what does 'plug and chug' refer to?
Heating a substance to observe phase changes
Mixing substances to observe reactions
Substituting known values into an equation
Using a calculator to solve equations
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If the problem asked for the heat in kilojoules, what would be the answer?
229.9 kJ
22.99 kJ
2.299 kJ
0.2299 kJ
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
