
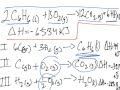
Enthalpy Change and Hess's Law
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of Hess's Law?
Determining the enthalpy change of a reaction
Balancing chemical equations
Measuring the temperature change in a reaction
Calculating the rate of reaction
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does flipping a reaction affect its enthalpy change?
It doubles the enthalpy change
It has no effect on the enthalpy change
It halves the enthalpy change
It changes the sign of the enthalpy change
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the example problem, what is the first step in using thermochemical equations?
Cancel out unnecessary components
Calculate the total enthalpy change
Identify the given equations
Balance the chemical equation
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the enthalpy change for the combustion reaction of C6H6?
-6534.8 kJ
49 kJ
-393.5 kJ
-285.8 kJ
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which component is multiplied by 12 in the detailed solution?
CO2
Oxygen
Water
Graphite
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final step in calculating the enthalpy change of a reaction?
Multiplying the enthalpy change by a factor
Flipping the reaction
Adding all equations together
Balancing the chemical equation
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many equations are used in the example problem to find the enthalpy change?
Five
Four
Two
Three
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
