
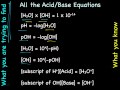
Acid-Base Chemistry Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary component of a solution that acts as the dissolving medium?
Molarity
Solute
Concentration
Solvent
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is concentration defined in terms of solute and solvent?
Mass of solute
Volume of solution
Amount of solvent in solute
Amount of solute in solvent
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does molarity measure in a solution?
Number of moles of solute per liter of solution
Volume of solvent
Concentration of solvent
Mass of solute
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which equation is used to determine the molarity of an acid from the concentration of H+ ions?
Subscript of base times concentration of base equals concentration of OH-
Concentration of OH- equals concentration of H+
Concentration of acid equals concentration of H+
Subscript of acid times concentration of acid equals concentration of H+
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the concentration of H3O+ ions in a 3 M solution of H2SO4?
3 M
9 M
1.5 M
6 M
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the pH of a solution given the concentration of H3O+ ions?
pH = log(concentration of H3O+)
pH = -log(concentration of H3O+)
pH = concentration of H3O+
pH = 1/concentration of H3O+
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the pOH of a solution if the concentration of OH- is 2 x 10^-15?
14
10
7
15
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
