
Properties and Structure of Water
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
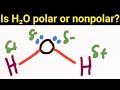
What is the conclusion about the polarity of water?
Water has no polarity.
Water's polarity depends on temperature.
Water is a polar molecule.
Water is a nonpolar molecule.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons does oxygen have in the Lewis structure of water?
2
4
6
8
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many bonds does oxygen form in a water molecule?
One
Four
Two
Three
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the hybridization of oxygen in a water molecule?
sp3
sp2
sp
sp3d
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the steric number of oxygen in water?
4
3
5
2
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to VSEPR theory, what is the shape of a water molecule?
Bent or V-shaped
Trigonal planar
Tetrahedral
Linear
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does the water molecule have a bent shape?
Due to the repulsion between lone pairs
Because of the hydrogen bonds
Due to the high temperature
Because of the ionic bonds
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
