
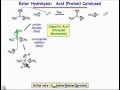
Proton-Catalyzed Reaction Mechanisms
Interactive Video
•
Chemistry
•
11th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the products of the ester hydrolysis reaction?
Carboxylic acid and ester
Ester and water
Carboxylic acid and methanol
Methanol and water
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of the zwitterionic intermediate in the uncatalyzed reaction?
It speeds up the reaction
It is the final product
It is a temporary charged species
It acts as a catalyst
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which step is considered the rate-determining step in the uncatalyzed reaction?
Formation of zwitterionic intermediate
β elimination
Proton transfer
Nucleophile addition
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the concentration of the key intermediate expressed in the rate law derivation?
Directly measured
Using fast equilibrium assumptions
Estimated from final products
Ignored in calculations
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What assumption is made about the backward reaction in the simplification process?
It is equal to the forward reaction
It is negligible
It is faster than the forward reaction
It is the main focus of the study
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the proton-catalyzed reaction, what role does the proton play?
It is the final product
It speeds up the rate-determining step
It is a reactant in the stoichiometry
It is consumed and not regenerated
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the proton affect the electrophile in the catalyzed reaction?
It weakens the electrophile
It converts the electrophile into a nucleophile
It makes the electrophile stronger
It has no effect
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
