
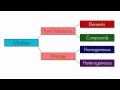
Classification of Matter Concepts
Interactive Video
•
Chemistry
•
6th - 8th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What was the initial classification of matter by the chemists?
Solids, liquids, and gases
Elements and compounds
Pure substances and mixtures
Homogeneous and heterogeneous
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the two main categories of matter discussed in the video?
Atoms and molecules
Solids and liquids
Elements and compounds
Pure substances and mixtures
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a characteristic of pure substances?
Definite and constant composition
Contains impurities
Variable composition
Always a mixture
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What example is used to explain a pure substance?
A jar of sand and sugar
A bag of salt
A piece of metal
A bottle of water
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is a mixture different from a pure substance?
It is a physical combination of pure substances
It has a constant composition
It is always homogeneous
It contains only one type of element
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What example is used to illustrate a mixture?
A piece of metal
A jar of sand and sugar
A bag of salt
A bottle of water
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the composition of a homogeneous mixture?
Varies in different parts
Uniform throughout
Always liquid
Always solid
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
