
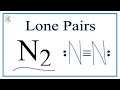
Lewis Structures and Nitrogen Bonds
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
6 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What percentage of the Earth's atmosphere is made up of nitrogen gas?
95%
50%
78%
21%
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are lone pairs in the context of chemical bonding?
Neutrons in the nucleus
Electrons involved in chemical bonds
Electrons not involved in chemical bonds
Protons in the nucleus
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many lone pairs are present in the Lewis structure of N2?
Three
One
Two
Four
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to draw the Lewis structure of N2?
To calculate the atomic mass
To identify the number of lone pairs
To determine the number of protons
To find the number of neutrons
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond is formed between the nitrogen atoms in N2?
Triple bond
Quadruple bond
Single bond
Double bond
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How are bonding pairs of electrons represented in a Lewis structure?
As triangles
As circles
As lines
As dots
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
