
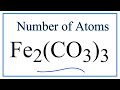
Chemical Composition and Carbonate Ions
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Ethan Morris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the chemical formula for Iron(III) Carbonate?
FeCO3
Fe2O3
Fe2(CO3)3
Fe3(CO3)2
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many iron atoms are present in Fe2(CO3)3?
2
4
3
1
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of carbon atoms in Fe2(CO3)3?
9
3
2
1
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many oxygen atoms are there in Fe2(CO3)3?
6
3
12
9
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of atoms in Fe2(CO3)3?
18
12
14
16
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the number of atoms in one mole of Fe2(CO3)3?
Add 6.022 x 10^23
Divide by 6.022 x 10^23
Multiply by 6.022 x 10^23
Subtract 6.022 x 10^23
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is Avogadro's number used for?
Measuring temperature
Calculating mass
Counting atoms in a mole
Determining volume
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
