
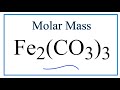
Molar Mass and Chemical Calculations
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Mia Campbell
FREE Resource
Read more
7 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of iron (Fe) as given in the video?
55.85 g/mol
12.01 g/mol
16.00 g/mol
291.73 g/mol
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many carbon atoms are present in the formula Fe2(CO3)3?
6
3
2
1
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of oxygen (O) as used in the calculation?
291.73 g/mol
55.85 g/mol
12.01 g/mol
16.00 g/mol
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why do we multiply the entire formula by three in the calculation?
Because there are three carbonate ions
Because there are three oxygen atoms
Because there are three iron atoms
Because there are three carbon atoms
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final calculated molar mass of iron(III) carbonate?
55.85 g/mol
12.01 g/mol
16.00 g/mol
291.73 g/mol
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What should you consider about your periodic table when calculating molar mass?
The size of the table
The rounding of decimal places
The number of elements
The color of the table
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If your periodic table rounds to more decimal places, what might happen to your answer?
It will be exactly the same
It will be significantly different
It will be incorrect
It might be slightly different
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
