
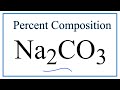
Percent Composition and Molar Mass
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main focus of the video tutorial?
Learning about the history of sodium carbonate
Understanding the chemical reactions of sodium carbonate
Finding the percent composition by mass of elements in Na2CO3
Calculating the density of sodium carbonate
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to calculate the molar mass of a compound?
To calculate the percent composition by mass
To measure its electrical conductivity
To determine its color
To find its boiling point
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of sodium carbonate (Na2CO3)?
16.00 grams per mole
44.01 grams per mole
22.99 grams per mole
105.99 grams per mole
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many sodium atoms are present in sodium carbonate?
One
Two
Three
Four
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the percent composition of sodium in sodium carbonate?
16.00%
43.38%
45.29%
11.33%
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the atomic mass of carbon used in the calculation?
12.01 grams per mole
16.00 grams per mole
22.99 grams per mole
105.99 grams per mole
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the percent composition of carbon in sodium carbonate?
43.38%
22.99%
11.33%
45.29%
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
