
Understanding Aqueous Ions and Acids
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
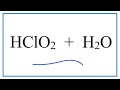
What is the primary indication that HClO2 is an acid?
The presence of H at the front
The presence of ClO2
Its color
Its reaction with metals
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is chlorous acid considered a weak acid?
It is not on the list of strong acids
It reacts violently with water
It completely dissociates in water
It has a high pH
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to chlorous acid in water?
It fully dissociates into ions
It partially dissociates into ions
It remains completely undissociated
It forms a solid precipitate
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the equilibrium arrow in the reaction indicate?
The reaction is exothermic
The reaction can proceed in both directions
The reaction is irreversible
The reaction goes to completion
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does 'aq' signify when written after ions?
The ions are in a liquid state
The ions are in a solid state
The ions are dissolved in water
The ions are in a gaseous state
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to note the aqueous state of ions?
It indicates the ions are reactive
It suggests the ions are volatile
It shows the ions are in a solution
It means the ions are stable
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is formed when H+ ions join with water molecules?
Oxygen molecules
Chloride ions
Hydronium ions
Hydroxide ions
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
