
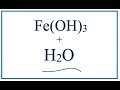
Iron(III) Hydroxide Solubility Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the expected outcome when iron(III) hydroxide is mixed with water?
It dissolves completely.
It forms a gas.
It reacts violently.
It dissociates into ions.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What ions are expected to form if iron(III) hydroxide dissociates in water?
Fe2+ and OH-
Fe3+ and OH-
Fe2+ and H+
Fe3+ and O2-
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the hydroxide ion formed during dissociation?
Variable
Positive
Negative
Neutral
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What coefficient is needed in front of OH- to balance the dissociation equation of iron(III) hydroxide?
4
3
1
2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to check a solubility table when predicting the behavior of iron(III) hydroxide in water?
To confirm the color change.
To determine the reaction speed.
To verify its solubility.
To measure the temperature change.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the solubility table indicate about iron(III) hydroxide?
It is insoluble.
It is slightly soluble.
It forms a precipitate.
It is highly soluble.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the 'I' symbol in the solubility table stand for?
Indeterminate
Ionic
Insoluble
Intense
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
